
ʵ��������NaNO3��������80 mL 1.4 mol��L-1��NaNO3��Һ���Իش�
(1)����������__________��
A.�ձ� B.500 mL����ƿ C.��Ͳ
D.��ͷ�ι� E.������ F.100 mL����ƿ
(2)����ʱӦ��ȡNaNO3__________g��
(3)ʵ�鿪ʼʱ����������ƿ__________��
(4)������һ�����ʵ���Ũ����Һ��ʵ���У����в�����������ҺŨ����Ӱ�����(д��ĸ) __________����ʹ������ҺŨ��ƫ�����__________����ʹ������ҺŨ��ƫС����__________��
A.���ձ����ܽ����ʣ�����ʱ��������������Һ
B.δ��ϴ���ձ��ڱڵ���Һת��������ƿ
C.����ƿ���������ҺҺ��δ���̶��߱�ֹͣ��ˮ
D.����õ���Һ������ƿת�Ƶ�����ྻ���Լ�ƿ��ʱ������������
E.���ձ�����Һת�Ƶ�����ƿ֮ǰ������ƿ������������ˮ
F.����ƿ��Һ�潫�ﵽ�̶���ʱ�����ӿ̶��ߺ�Һ��
(1) B (2) 11.9 (3) �Ƿ�©ˮ (4) D��E C��F A��B
���������������1������û��80ml������ƿ������Ӧ������100ml������Ҫ��������500ml����ƿ����ѡB��
��2������ʱӦ��ȡ�����ƹ����������0.1L��1.4mol/L��85g/mol��11.9g��
(3)ʵ�鿪ʼʱ����������ƿ�Ƿ�©ˮ��
��4������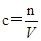 ��֪�����ձ����ܽ����ʣ�����ʱ��������������Һ�������ʵ��������٣�Ũ��ƫ�ͣ�δ��ϴ���ձ��ڱڵ���Һת��������ƿ��ͬ���������������٣�Ũ��ƫ�ͣ�����ƿ���������ҺҺ��δ���̶��߱�ֹͣ��ˮ��������ƿ����Һ������٣�Ũ��ƫ�ߣ�����õ���Һ������ƿת�Ƶ�����ྻ���Լ�ƿ��ʱ��������������Ũ�Ȳ��䣻���ձ�����Һת�Ƶ�����ƿ֮ǰ������ƿ������������ˮ������Ӱ��ʵ����������ƿ��Һ�潫�ﵽ�̶���ʱ�����ӿ̶��ߺ�Һ�棬������ƿ����Һ������٣�Ũ��ƫ�ߡ�
��֪�����ձ����ܽ����ʣ�����ʱ��������������Һ�������ʵ��������٣�Ũ��ƫ�ͣ�δ��ϴ���ձ��ڱڵ���Һת��������ƿ��ͬ���������������٣�Ũ��ƫ�ͣ�����ƿ���������ҺҺ��δ���̶��߱�ֹͣ��ˮ��������ƿ����Һ������٣�Ũ��ƫ�ߣ�����õ���Һ������ƿת�Ƶ�����ྻ���Լ�ƿ��ʱ��������������Ũ�Ȳ��䣻���ձ�����Һת�Ƶ�����ƿ֮ǰ������ƿ������������ˮ������Ӱ��ʵ����������ƿ��Һ�潫�ﵽ�̶���ʱ�����ӿ̶��ߺ�Һ�棬������ƿ����Һ������٣�Ũ��ƫ�ߡ�
���㣺����������ѡ��ʵ����������ʵ���Ũ�ȵ����ơ������Լ�������
�����������Ǹ߿��еij������ͣ������е��Ѷ�����Ŀ��飬���������ǿ�����ض�ѧ������֪ʶ�Ĺ��̺�ѵ������Ҫ�ǿ���ѧ��������û���֪ʶ���ʵ�����������������������ѧ����ʵ���������������������Ҫ���Գ���������ѡ�á�ʵ���������Ϊ���ģ�ͨ����ʲô��Ϊʲô���������ص㿼��ʵ����������Ĺ淶�Ժ�ȷ�Լ��������֪ʶ���ʵ�������������������ѵ���������������cB��nB/V�ɵã�һ�����ʵ���Ũ����Һ���Ƶ����������ʵ����ʵ�����B����Һ�����V����ġ�������ʱ���ؼ�Ҫ�����ƹ�������������V�����ı仯��������һ�����ʵ���Ũ����Һʱ����nB������ֵС����V������ֵ��ʱ������ʹ������ҺŨ��ƫС����nB������ֵ��V������ֵСʱ������ʹ������ҺŨ��ƫ�ݴ��жϡ�


 ��У����ϵ�д�
��У����ϵ�д�
| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
ʵ����������80mL 1.4mol/L��NaNO3��Һ���Իش�
��1���ò����������ǣ� ��
����A.�ձ� B.500mL����ƿ C.��Ͳ D.��ͷ�ι� E.������ F.100mL����ƿ
��2������ʱӦ��ȡNaNO3 g��
��3�����в�����˳���ǣ�����ĸ��ʾ�� ��
����A.��ȴ B.���� C.ϴ�� D.���� E.�ܽ� F.ҡ�� G.ת��
��4��������һ�����ʵ���Ũ����Һ��ʵ���У����в������������Һ��Ӱ����ǣ�д��ţ� ����ʹ������Һ��Ũ��ƫ����� ��
A�����ձ����ܽ����ʣ�����ʱ��������������Һ��
B��δ��ϴ���ձ��ڱڵ���Һת��������ƿ��
C������ƿ���������ҺҺ��δ���̶��߱�ֹͣ��ˮ��
D������õ���Һ������ƿת�Ƶ�����ྻ���Լ�ƿ��ʱ��������������
E�����ձ�����Һת�Ƶ�����ƿ֮ǰ������ƿ������������ˮ��
F��������ƿ��Һ�潫�ﵽ�̶���ʱ�����ӿ̶��ߺ�Һ�档
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
��6�֣�ʵ����������80mL 1.4mol/L��NaNO3��Һ���Իش�
��1���ò����������ǣ� ��
�� A.�ձ� B.500mL����ƿ C.Բ����ƿ D.��ͷ�ι� E.������
��2������ʱӦ��ȡNaNO3 g��
��3��������һ�����ʵ���Ũ����Һ��ʵ���У����в�����ʹ������Һ��Ũ��ƫ����� ��
A�����ձ����ܽ����ʣ�����ʱ��������������Һ��
B��δ��ϴ���ձ��ڱڵ���Һת��������ƿ��
C������NaNO3����ʱ��ƽָ��ƫ����
D������õ���Һ������ƿת�Ƶ�����ྻ���Լ�ƿ��ʱ��������������
E�����ձ�����Һת�Ƶ�����ƿ֮ǰ������ƿ������������ˮ��
F������ʱ����Һ��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ��2014��ɽ��ʡ�߶���ѧ�����п��Ի�ѧ�Ծ��������棩 ���ͣ������
ʵ��������NaNO3��������80 mL 1.4 mol��L-1��NaNO3��Һ���Իش�
(1)����������__________��
A.�ձ� B.500 mL����ƿ C.��Ͳ
D.��ͷ�ι� E.������ F.100 mL����ƿ
(2)����ʱӦ��ȡNaNO3__________g��
(3)ʵ�鿪ʼʱ����������ƿ__________��
(4)������һ�����ʵ���Ũ����Һ��ʵ���У����в�����������ҺŨ����Ӱ�����(д��ĸ) __________����ʹ������ҺŨ��ƫ�����__________����ʹ������ҺŨ��ƫС����__________��
A.���ձ����ܽ����ʣ�����ʱ��������������Һ
B.δ��ϴ���ձ��ڱڵ���Һת��������ƿ
C.����ƿ���������ҺҺ��δ���̶��߱�ֹͣ��ˮ
D.����õ���Һ������ƿת�Ƶ�����ྻ���Լ�ƿ��ʱ������������
E.���ձ�����Һת�Ƶ�����ƿ֮ǰ������ƿ������������ˮ
F.����ƿ��Һ�潫�ﵽ�̶���ʱ�����ӿ̶��ߺ�Һ��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ��2013�콭��ʡ�����н����Ҹ�һ��ѧ����ĩ���Ի�ѧ�Ծ� ���ͣ�ʵ����
��6�֣�ʵ����������80mL 1.4mol/L��NaNO3��Һ���Իش�
��1���ò����������ǣ� ��
�� A.�ձ� B.500mL����ƿ C.Բ����ƿ D.��ͷ�ι� E.������
��2������ʱӦ��ȡNaNO3 g��
��3��������һ�����ʵ���Ũ����Һ��ʵ���У����в�����ʹ������Һ��Ũ��ƫ����� ��
A�����ձ����ܽ����ʣ�����ʱ��������������Һ��
B��δ��ϴ���ձ��ڱڵ���Һת��������ƿ��
C������NaNO3����ʱ��ƽָ��ƫ����
D������õ���Һ������ƿת�Ƶ�����ྻ���Լ�ƿ��ʱ��������������
E�����ձ�����Һת�Ƶ�����ƿ֮ǰ������ƿ������������ˮ��
F������ʱ����Һ��
�鿴�𰸺ͽ���>>
����ѧУ��ѡ - ��ϰ���б� - �����б�
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com