
��4 �֣���ֽ���ҹ��Ŵ��Ĵ���֮һ������Ч���ƶ������������ķ�չ��
�Ż���ũ����Ľո���Ϊ��ֽԭ�ϣ����Լ��ٷ��սոѴ������̳���Ⱦ���ոѵ���Ҫ �ɷ�֮һ����ά��[��ѧʽ (C6H10O5)n]������������������Ԫ����______�� ��ά������______��ѡ��л����������������֮һ����
����ֽ�����л���������� NaOH �ķ�ˮ��Сӱͬѧ���� pH ��ֽ���Բⶨ��ˮ���ȣ��ⶨ�ľ��巽���� ��
��Ϊ�����÷�ˮ�Ի�������Ⱦ�����ϡ���ᡢ����ͭ��̼�������ҩƷ��ѡ��һ��ҩƷ������ȥ��ˮ�е�NaOH������д���ó��ӷ�Ӧ�Ļ�ѧ����ʽ�� ��
�� ��Ԫ�� �л��� �� 2NaOH + H2SO4 = Na2SO4 + 2H2O
�� ��pH��ֽ���ڽྻ����İ״�Ƭ�������ϣ��ýྻ����IJ�����պȡ����Һ��
����pH��ֽ�ϣ�Ѹ�������ɫ�����ж��ա�
���������������1������ά���У���Ԫ�ص����ԭ��������࣬�������������ά�����к���̼Ԫ�����л��
��2���ⶨ��ҺPH�ķ������ò�����պȡ��������Һ����PH��ֽ�ϣ��ٽ���ʾ����ɫ�����ɫ�����ж��գ�
��3��ϡ���ᡢ����ͭ��̼�������ҩƷ��ֻ��ϡ���������������Ʒ����кͷ�Ӧ��2NaOH + H2SO4 = Na2SO4 + 2H2O
���㣺�й���Է��������ļ��㡢PH�IJⶨ���кͷ�Ӧ


 Сѧ�̲�ȫ��ϵ�д�
Сѧ�̲�ȫ��ϵ�д� Сѧ��ѧ������ѿڶ���ϵ�д�
Сѧ��ѧ������ѿڶ���ϵ�д� ������Ӧ�������������ϵ�д�
������Ӧ�������������ϵ�д� �㽭֮�ǿ�ʱ�Ż���ҵϵ�д�
�㽭֮�ǿ�ʱ�Ż���ҵϵ�д�
| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ���Ϣ������
ij������Һ�к����Ȼ��ơ��Ȼ�ͭ���Ȼ�������ij����С�����÷�Һ��ȡ����ͭ��Cu�������������ֲ�Ʒ����������ͼ��ʾ�������в���������Ԫ�أ���
��ش�
��1���Լ�A�� ������B�ijɷ��� �� ��ƷG�� ��
��2������C�����ķ����� ��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
��6�֣�ũ����������������������߲�Ʒ�IJ�����������ij���ӵ���������Ӫ��Һ�к���7��08%��KNO3��
��1��KNO3�е�Ԫ�ص����������� ���������һλС������KNO3���� ��ѡ����ʡ������طʡ� �����ʡ����Ϸ��ϡ�)��
��2��ũҵ��������35��4%��KNO3��Һ������150kg��Ӫ��Һ����Ҫˮ������ kg��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
����̼�����ơ������ǡ�������������������ʣ�ѡ����ʵ������û�ѧʽ��գ�
��1��������Ǧ�����к��е��� ��
��2����Ϊ���幩�ܵ������� ��
��3�����Ƹ��ķ��ͷ۳ɷ�֮һ���� ��
��4������������θ�����ļ��� ��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
(10��)�Ϻ������㵺���丽���������ҹ������������̲��ŷḻ�ĺ�����Դ��
��1����ˮ���Ρ��Ӻ�ˮ��ȡ�Ĵ����к�����ɳ������þ���Ȼ��Ƶ����ʣ�Ϊ�õ��ϴ����Ȼ��ƣ�����������ˮ��Ȼ��������²�����a���ӹ�����Ba(OH)2��Һ��b�����Թ��������c���ӹ�����Na2CO3��Һ��d�����ˣ�e����������ȷ�IJ���˳���� (����ĸ)����Na2CO3��Һ�������dz�ȥ ��
��2����ˮ��������ͼ��һ��������������ˮ��װ�á�֤���õ���ˮ�ǵ�ˮ�ķ����� ��
��3����ˮ��þ���Ӻ�ˮ����ȡ����þ�Ĺ�������ͼ��ʾ��
������ת�������з����кͷ�Ӧ���� (�Ӧ˳���)����ˮ�б����ͺ����Ȼ�þ����١��������������� ��
��4����ˮ���Ƽ������ƴ�������ͨ����ѧ��Ӧ����NaHCO3��NH4Cl��NaHCO3�ᾧ�������ټ���NaHCO3�Ƶô��
�ٰ���ƴ���������NaHCO3��NH4Cl�Ļ�ѧ����ʽΪ ��
�ڷ�Ӧ������NaHCO3��NH4Cl��������Ϊ84��53��5��NaHCO3��NH4Cl���ܽ��������ͼ��ʾ�������NaHCO3�ᾧ������NH4Clû�нᾧ������ԭ���� ��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
(7��)�ƵĻ��������ճ�������Ӧ�ù㷺��
��1��̼�������Ƿ��ͷ۵ijɷ�֮һ�����׳��� ��
��2���������ƣ�NaClO����84����Һ����Ч�ɷ֣�������������Ԫ�صĻ��ϼ�Ϊ ��
��3���Ȼ�������Ҫ�ĵ�ζƷ����������ʾ��ͼ�ֱ��ʾ��ͬ�Ļ�ѧ��Ӧ�����������ж����Ȼ��ơ�

ͼ1 ͼ2
ͼ1������������Ӧ�����Ȼ��Ƶ�ʾ��ͼ���Ȼ������� ��ѡ�ԭ�ӡ���
�����ӡ������ӡ������ɵģ��ɴ˿�����Ԫ�صĻ�ѧ������ ������ء�
ͼ2����������������Һ�����ᷴӦ����ʵ�ʣ��÷�Ӧ�Ļ�����Ӧ����Ϊ ��
��4�� �������ƣ�NaN3�����㷺Ӧ����������ȫ���ң�������ײ����30����������NaN3Ѹ�ٷֽ��������ֵ��ʣ���Ӧ�Ļ�ѧ����ʽΪ ��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
Ϊ���ѧѧ����ȡ�õijɾ��Լ������������Ĺ��ף����Ϲ���2011�궨Ϊ�����ʻ�ѧ�ꡱ����ش��������⣺
��1�����ϵķ�չ�ƶ����Ľ��������в������ںϳɲ��ϵ��� ������ţ�
| A������ϩ���� | B������� | C���� | D����ë |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
(6��)�����ܽ�Աȷ�����ѧϰ��ѧ���õĿ�ѧ��������ͼ��ʾ����Բ�ص��������ʾ���ǵĹ�ͬ���������Ƶ㣩���ص���������IJ��ֱ�ʾ���ǵĶ�����������ͬ�㣩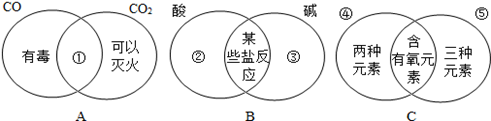
��1���۲�Aͼ������ɽǶ�д������ͼ��������Ӧ���ʵ�����
������һ�㣩
��2���۲�Bͼ��д�����Ϣڵ���Ļ�ѧ����
�� ���������㣩���ɿ�����ͼ����ijЩ�η�����Ӧ������д�����������ᷴӦ����������ʯ�ҷ�����ѧ��Ӧһ���εĻ�ѧʽΪ ��
��3���۲�Cͼ�������ʷ�����д�����Ϣܢݵ����ʵĻ�ѧʽ �� ��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
��4�֣�
��ϪѧУ�Ļ���С���������о�ֽ��������ˮԴ��Ⱦ״������������ʵ�飺
��1������Ϊ����Ʒ���ʱ�ⶨ��ˮ���ɼ���ӦΪ ��
A�����ξ�������ˮ�� B�����ξ�������ˮ��
C����ֽ�����۴� D��������ѡ
��2��������ֽ������Ϫ�����ŷŷ�Һ��pHΪ8�����Һ�� �ԣ�����ˮ�в����ܺ��д����������� ��
A�� H+ B��OH- C��Na+ D��CO32-
��3����������з����жϷ�Һ������ԣ��ó��Ľ����д������ ��
A��������ɫ��̪��Һ����Һ���ɫ����÷�Һ�Լ���
B��������ɫʯ����Һ����Һ����ɫ����÷�Һ�Լ���
C��ͨ�������̼���壬��Һ�����Ա仯����÷�Һ�Լ���
D�����Թ�ȡ��Һ��������������ͭ��Һ������ɫ�������ɣ���÷�Һ�Լ���
�鿴�𰸺ͽ���>>
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com