���ž��õķ�չ����Դ�ͻ��������Ϊ���ǹ�ע�Ľ��㣮
��1��Ŀǰ�������Ի�ʯȼ��Ϊ��Ҫ��Դ�������Ļ�ʯȼ�ϰ���ú��
����Ȼ������Ȼ��ȼ�յĻ�ѧ����ʽΪ
��
��2��Ϊ��ʹú���ȼ�գ�ͨ����ú������ú�ۺ�ȼ�գ���Ŀ����
��
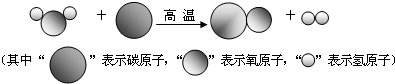
��3��Ϊ�����������������̼���ŷţ���ѧ�ҽ�������̼�������ڴ����ͼ��ȵ������·�Ӧ��ת��Ϊˮ�ͼ��飬�÷�Ӧ�Ļ�ѧ����ʽΪ
��
��4�������������Դ��������������������������Ҫ�����������ܡ����ܡ�
����һ�֣�������Դ��
��5��2013��4��2�գ�������Ȼ��������й�������V�ͣ�Green Week���ж��������빫�ڴ�С�����𣬼��л�����������Ϊ�У����ϡ���V�͡���������
��
A�������������°� B�����û���һ����ľ��
C������ʹ�û�������� D�����û���һ����ֽ����