


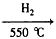 FexNy+ H2��δ��ƽ���������İ���34.0 g�����ij�����ʯ
FexNy+ H2��δ��ƽ���������İ���34.0 g�����ij�����ʯ ��������һ���þ�ϵ�д�
��������һ���þ�ϵ�д� Сѧ��10����Ӧ����ϵ�д�
Сѧ��10����Ӧ����ϵ�д�
| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ��Ķ�����
| O | 2- 7 |
| O | 2- 4 |
| O | 2- 7 |

| ������ | Al��OH��3 | Fe��OH��3 | Fe��OH��2 | Cu��OH��2 |
| pH | 5.2 | 3.2 | 9.7 | 6.7 |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ��2013-2014ѧ�꽭��ʡ�Ͼ��С������и����ڶ���ģ�⿼�Ի�ѧ�Ծ��������棩 ���ͣ������
CoxFe3��xO4�ŷ���һ�ֱȽϺõĸ߽������ŷۡ���ҵ����FeSO4Ϊԭ���Ʊ�CoxFe3��xO4����Ҫ�������£�

��1������������FeSO4��Һ�м���NaOH��Һ����40���½�������FeOOH���֡����ɾ��ֵĻ�ѧ����ʽΪ?????? ��
��2���������������Ƶ�����FeSO4��Һ����Ƥ���������У����µ�60������������������ֳ���һ���ߴ���ˡ�ˮϴ�������FeOOH��ĩ���������з�����Ƥ��Ŀ����?????? ���������������Ϊ??? ??? ��
��3����������FeOOH��200��300���¼�����ˮ�����ɺ�ɫFe2O3��ʵ������ɸò�����Ҫ���������е�??? ??? (����ĸ)��
a��������b���ձ�����c����������d�������ǡ���e���ƾ���
��4��������ͨ��H2��������300��400��������Fe3O4��ͨ��H2ǰҪ�����¯��ͨ��N2��������Ϊ??? ??? ��
��5������������CoSO4��Һ�����ôֲ�Ʒ�����ˡ�ϴ�ӡ����T�ó�Ʒ������ֲ�Ʒϴ�Ӹɾ���ʵ�������������???????? ��
��6��ij�о�С����������ӵ����������(��LiCoO2������������������)�Ʊ�CoSO4��7H2O���塣�±��г�����ؽ������������������������pH(��ʼ������pH����������Ũ��Ϊ1.0 mol��L��1����)��
�������� | ��ʼ������pH | ������ȫ��pH |
Fe3�� | 1.1 | 3.2 |
Fe2�� | 5.8 | 8.8 |
Co2�� | 6.9 | 9.4 |
���������ʵ�鲽��(��ѡ�õ��Լ���H2O2��ϡ���ᡢϡ���ᡢNaOH��Һ)��
����N����������ͪ��120���½�ϴ�������ϣ�ʹLiCoO2���������룬�õ�LiCoO2��Ʒ����������
��???????? ��
�������ô�ƷCoSO4��Һ�м���NaOH��Һ������pHԼΪ5�����ˡ�
��???????? ��
����Co(OH)2��������ϡ�����У�����Ũ�������½ᾧ���õ�CoSO4��7H2O���塣
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ��2012-2013ѧ��ӱ�ʡ�����и����ڶ���ģ�⿼�����ۻ�ѧ�Ծ��������棩 ���ͣ�������
���ǵؿ��к����ڶ��Ľ���Ԫ�أ��䵥�ʡ��Ͻ����������������е�Ӧ�ù㷺��
(�D)��ҵ��ˮ�к���һ������Cr2O72����CrO42�������ǻ�����༰��̬ϵͳ�����ܴ��Σ����������д��������õĴ��������ǵ�ⷨ���÷���Fe���缫��⺬Cr2O72�������Է�ˮ�����ŵ��Ľ��У�����������ҺpH���ߣ�����Cr��OH��3������
��1����Fe���缫��Ŀ���� ��
��2������������ҺpH���ߵ�ԭ����_ (�õ缫��Ӧʽ���ͣ�����Һ��ͬʱ���ɵij������� ��
(��)�������ŷ���һ�ִż�¼���ϣ����ð�����400�����Ϸֽ�õ��ĵ�ԭ�������ߴ������п��Ʊ����������Ʊ��ߴ������漰����Ҫ�����������£�
��֪��ij������ʯ��60.0%Fe2O3��3.6%FeO��������Al2O3��MnO2��CuO�ȡ�
�ڲ���������������������ʽ��ȫ����ʱ��Һ��pH���£�

��3��������м�˫��ˮ��Ŀ����
��pH������3.4��������
����֪25��ʱ��Ksp[Cu��OH��2] =2.0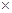 10-20�����¶��·�Ӧ��Cu2����2H2O
10-20�����¶��·�Ӧ��Cu2����2H2O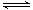 Cu(OH)2��2H����ƽ�ⳣ��K=
��
Cu(OH)2��2H����ƽ�ⳣ��K=
��
��4������жϲ�����г����Ƿ�ϴ�Ӹɾ��� ��
��5���Ʊ��������ŷ۵ķ�Ӧ��Fe��NH3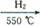 FexNy��H2 (δ��ƽ��,���������������İ���34.0g�����ij�����ʯ2 kg������������������ģ���FexNy�ŷ۵Ļ�ѧʽΪ
��
FexNy��H2 (δ��ƽ��,���������������İ���34.0g�����ij�����ʯ2 kg������������������ģ���FexNy�ŷ۵Ļ�ѧʽΪ
��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ��2012-2013ѧ��ɽ��ʡ̩���и��������ָ�ϰ����������ۻ�ѧ�Ծ��������棩 ���ͣ������
���ǵؿ��к����ڶ��Ľ���Ԫ�أ��䵥�ʡ��Ͻ��������������е�Ӧ�ù㷺��
��һ����ҵ��ˮ����һ������Cr2O72--��CrO42-�����ǻ�����༰��̬ϵͳ�����ܴ��Σ����������д��������õĴ��������ǵ�ⷨ���÷���Fe���缫��⺬Cr2O72-�����Է�ˮ�����ŵ��Ľ��У�����������ҺpH���ߣ�����Cr(OH)3������
��1����Fe���缫��Ŀ����____________________________________________________��
��2������������ҺpH���ߵ�ԭ����___________���õ缫��Ӧʽ���ͣ�����Һ��ͬʱ���ɵij�������___________��
�������������ŷ���һ�ִż�¼���ϣ����ð�����400�����Ϸֽ�õ��ĵ�ԭ�������ߴ������п��Ʊ����������Ʊ��ߴ����漰����Ҫ�����������£�
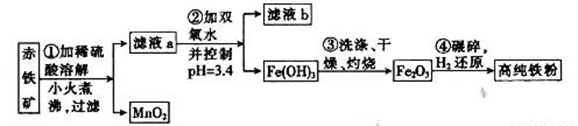
��֪����ij������ʯ��60.0 Fe2O3��3.6% FeO��������Al2O3��MnO2��CuO�ȡ�
Fe2O3��3.6% FeO��������Al2O3��MnO2��CuO�ȡ�
�ڲ���������������������ʽ��ȫ����ʱ��Һ��pH��

��3��������м�˫��ˮ��Ŀ����__________________��pH������3.4��������_______________________����֪25��ʱ��Ksp[Cu(OH) 2] =2.0��10-20�����¶��·�Ӧ��Cu2++2H2O Cu(OH) 2+2H+��ƽ�ⳣ��K=_______��
Cu(OH) 2+2H+��ƽ�ⳣ��K=_______��
��4������жϲ�����г����Ƿ�ϴ�Ӹɾ���_____________________________________��
�鿴�𰸺ͽ���>>
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com