
18-�� ���������������ڵ������͡�����ܵ�ʳ���㾫����ϳɷ�Ӧ�Ļ�ѧ����ʽ���£�
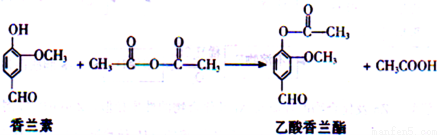

����������ȷ���ǣ�_____��
A���÷�Ӧ����ȡ����Ӧ
B�������������ķ���ʽΪC10H8O4
C��FeCl3��Һ����������������������������
D������������������NaOH��Һ��ˮ��õ������������
18-���� A��CH2=CH2���� D(HOOCCH=CHCH=CHCOOH)�ϳɸ߷���P����ϳ�·�����£�
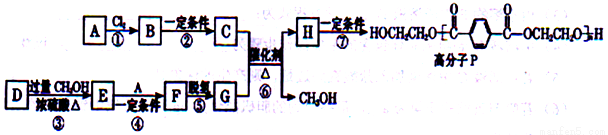
��֪����
�����봼�ɷ���������������Ӧ��

��1��B������Ϊ_________________��D�й����ŵ�����Ϊ_________________________��
��2��C�ķ���ʽ��C2H6O2����Ӧ�ڵ��Լ��ͷ�Ӧ������____________________________��
��3��F�Ľṹ��ʽ��__________________________��
��4����Ӧ�Ļ�ѧ����ʽ��____________________________________��
��5��G��һ��ͬ���칹��G'Ϊ���������˴Ź���������3�ַ���1mol���л�������������ˮ���������2 mol NaOH��Ӧ��G'�Ľṹ��ʽΪ___________________��
��6���ԶԱ����״����״�Ϊ��ʼԭ�ϣ�ѡ�ñ�Ҫ�����Լ��ϳ�G��д���ϳ�·�ߣ��ýṹ��ʽ��ʾ�л���ü�ͷ��ʾת����ϵ����ͷ��ע���Լ��ͷ�Ӧ��������___________________

| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ��2016-2017ѧ��ӱ�ʡ�߶���ѧ�ڵ�һ���¿���ѧ�Ծ��������棩 ���ͣ�ѡ����
��NAΪ�����ӵ�������ֵ������˵����ȷ���� ( )
A�����³�ѹ�£�16 g ��O2��O3��ɵĻ����������������Ϊ8NA
B��ʵ���ҷֱ���KClO3��H2O2��ȡ3.2g O2ʱ��ת�Ƶĵĵ�������Ϊ0.4NA
C��25��ʱ��1 L pH��1��ϡ������Լ��2NA��H+
D����״���£�22.4 L SO3����������ΪNA
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ��2017������и�����ѧ�ڵ�һ���¿���ѧ�Ծ��������棩 ���ͣ������
�����ʾΪԪ�����ڱ���һ���֣�����Ԫ�آ١����ڱ��е�λ�ã���ش��������⣺
�� ���� | IA | 0 | ||||||
1 | �� | ��A | ��A | ��A | ��A | ��A | ��A | |
2 | �� | �� | �� | �� | ||||
3 | �� | �� | �� | �� | ||||
��1���ۡ��ܡ��ߵ�ԭ�Ӱ뾶�ɴ�С��˳����_________����Ԫ�ط��ű�ʾ����
��2��������ʵ��˵����Ԫ�صķǽ����ԱȢ�Ԫ�صķǽ�����ǿ����__________��
a.�ڵĵ������Ԫ�صļ��⻯����Һ��Ӧ����Һ�����
b.��������ԭ��Ӧ�У�1mol�ڵ��ʱ�1mol���ʵõ��Ӷ�
c.�ں͢���Ԫ�صļ��⻯�����ȷֽ⣬ǰ�ߵķֽ��¶ȸߡ�
��3���١�������Ԫ�ذ�ԭ�Ӹ���֮��Ϊ1��1��ɵij���Һ̬�������������Һ���ܽ�Fe2+ ������д���÷�Ӧ�����ӷ���ʽ ___________________��
��4�� ��֪���ڱ��д��ڶԽ����ƹ������루Be��������ѧ�������ƣ�����������������Ҳ�����ԣ�д���������������ܵ�����������ˮ���ﷴӦ�Ļ�ѧ����ʽ _______________________��
��5����֪W+X=Y+Z����Ӧ��Ҫ���ȣ�����W��X��Y��Z�ֱ����ɢ٢ڢ�����Ԫ���γɵ�����10�������ӣ�W��XΪ���ӣ�Y��ZΪ���ӣ���д���û�ѧ����ʽ_________________��
��6���ɱ���Ԫ���γɵ����ʿɷ�����ͼ�еķ�Ӧ������B��C��G�ǵ��ʣ�BΪ����ɫ���壬 D��Һ�Լ��ԡ�
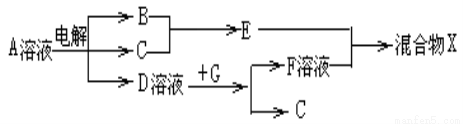
��д��D��Һ��G��Ӧ�����ӷ���ʽ______________________��
��д������A��Һ�����ʵ������ӵķ���____________________��
�۳����£������1L0.1mol/L��A��Һ��һ��ʱ�������ҺpHΪ12��������Һ����仯������õ�������ת�Ƶ��ӵ����ʵ���Ϊ��________________��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ��2016-2017ѧ��ɽ��ʡ��һ3���¿���ѧ�Ծ��������棩 ���ͣ�ѡ����
Ԫ��R��X��T��Z��Q��Ԫ�����ڱ��е����λ�����±���ʾ,����R�����ڰ�����H2���һ��ϲ�������ը���������ж���ȷ����(����)
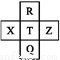
�ٷǽ�����:Z<T<X ��R��Q�ĵ��������26
����̬�⻯���ȶ���:R<T<Q ������������ˮ���������:T>Q
A. �٢� B. �٢� C. �ڢ� D. �ڢ�
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ��2016-2017ѧ��ɽ��ʡ��һ3���¿���ѧ�Ծ��������棩 ���ͣ�ѡ����
���й��ڼ����Ԫ�غ�±�ص�˵���д������(����)
A. ��˵����������,�����Ԫ�غ�±�ص�ԭ�Ӱ뾶��������
B. �����Ԫ����,�ԭ��ʧȥ�������ӵ���������;±����,��ԭ�ӵõ���������ǿ
C. �嵥����ˮ�ķ�Ӧ���ȵ�����ˮ�ķ�Ӧ������
D. ����ˮ�ķ�Ӧ������ˮ�ķ�Ӧ������
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ʡ���IJ���ѧ2017�������ѧ��������ѧ�Ծ� ���ͣ������
A��B��C��D��E���ֶ���������Ԫ�أ�ԭ������������������C��D��Eͬ���ڣ�A��Cͬ���壬B��Eͬ���壬BԪ�ص�ԭ�������������Ǵ�������������������֪A�������ܶ���С�����塣
��ش��������⣺
��1��Ԫ��C�����ڱ��е�λ��______________________________��
��2�� A��C��E��ԭ�Ӹ�����1��1��1�γɻ�����X�������ʽΪ_________________��
��3�� B��E��Ӧ���⻯���ȶ��ԵĴ�С˳���ǣ��÷���ʽ��ʾ�� ________________��
��4����D�Ƿǽ���Ԫ�أ��䵥���ڵ��ӹ�ҵ������ҪӦ�ã���д��������������ǿ����Һ�����ӷ���ʽ��___________________________________________��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ʡ���IJ���ѧ2017�������ѧ��������ѧ�Ծ� ���ͣ�ѡ����
����װ�û�ʵ�������ȷ����

A. ����pH��ֽ��ij��Һ������� B. ��̽�������ԣ�KMnO4>Cl2>I2
C. �����հ����ư�ˮ D. ���к͵ζ�ʵ��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ��2016-2017ѧ���Ĵ�ʡ�ɶ��б���У���߶�3���¿���ѧ�Ծ��������棩 ���ͣ�ѡ����
�����뻯ѧ�����йص�˵����ѧ���ﲻ��ȷ���ǣ� ��
A. ��ʯī���缫���NH4Cl��Һ��������ӦʽΪ��2Cl����2e����Cl2��
B. ��Al��Cu��Ũ������ɵ�ԭ��أ���������ӦʽΪ��NO3����2H����e��= NO2����H2O
C. ��Al��Mg��NaOH��ɵ�ԭ��أ��为����ӦʽΪ��Al��3e��+3OH�� = Al(OH)3��
D. Mg(OH)2��������Һ�д����ܽ�ƽ�⣺ Mg(OH)2(s)  Mg2+(aq) + 2OH��(aq)���ù��������NH4Cl��Һ
Mg2+(aq) + 2OH��(aq)���ù��������NH4Cl��Һ
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ��2017���㽭ʡ����������ȫ������3��������ѡ����Ŀ����ѧ�Ծ��������棩 ���ͣ������
��������H2��ԭCuCl�Ʊ�����ͭ����Ӧԭ�����£�
2Cu(s)+Cl2 (g) 2CuCl(s) ��H1=-36 kJ��mol-1 ��
2CuCl(s) ��H1=-36 kJ��mol-1 ��
H2(g)+2CuCl( s)=2Cu(s)+2HCl(g) ��H2 ��
�й����ʵļ����������±���
���� | H2 | Cl2 | HCl |
����(kJ��mol-1) | 436 | 243 | 432 |
(1)���H2=_______ kJ��mol-1��
(2)���ⶨ��Ӧ���Ʊ�����ͭ�ķ�Ӧ���ƴ�ԭ����_______________��
(3)��ij�¶��£���Ӧ�ٴﵽƽ��״̬����tlʱ������ѹǿ��ԭ����2����Cu�����㹻������ͼ�л���Cl2Ũ�ȵı仯�����ߡ�_______________
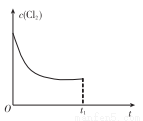
(4)��ɫ������ˮ��CuCl�����ɵ�ⷨ�Ƶã���ͼ��ʾ��
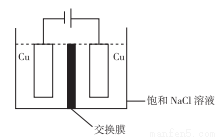
��װ�����õĽ���ĤΪ_____________��
A.�����ӽ���Ĥ B�������ӽ���Ĥ C�����ӽ���Ĥ D�����������ӽ���Ĥ
�������ĵ缫��ӦʽΪ_________________��
(5)��֪CuCl���ܽ���ϡ���ᣬд���÷�Ӧ�Ļ�ѧ����ʽ��_________________��
(6)������ѧ֪ʶд����ȡCuCl��һ�ַ������û�ѧ����ʽ��ʾ��_________________��
�鿴�𰸺ͽ���>>
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com