
��1������������ѡ�������������ñ����ĸ��д��__________��
A.������ƽ�������룩 B.�в� C.�Թܼ� D.�ƾ��� E.������ F.������ G.���� H.������ I.ʯ���� J.���ż�?
�����������⣬����Ҫ��������__________��
��2��ijѧ��ʵ���õ��������ݣ�
����ǰ���� | ���Ⱥ����� | |
W1�������� | W2������+���壩 | W3������+��ˮ����ͭ�� |
5.4 g | 7.9 g | 6.8 g |
��д���ᾧˮ������x%���ļ��㹫ʽ����W1��W2��W3��ʾ��
x%=__________�������ⶨ�����ƫ����ƫ�ͣ�__________��
�����з�����ѡ����ѧ������ʵ������ԭ������ǣ���д��ĸ��__________��
A.����ǰ����ʱ����δ��ȫ����
B.������μ��Ⱥ���������ϴ���0.1 g)
C.���Ⱥ�����δ�������������ȴ
D.���ȹ����о�����������ʧ
 �����Ļ���������人������ϵ�д�
�����Ļ���������人������ϵ�д�
| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ��Ķ�����
| 0.14CV |
| m |
| 14CV |
| m |
| 0.14CV |
| m |
| 14CV |
| m |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ��Ķ�����
 ʵ������Ũ������ͭ�ķ�Ӧ��ȡ���� NaHSO3��ʵ��װ����ͼ��ʾ����ش�
ʵ������Ũ������ͭ�ķ�Ӧ��ȡ���� NaHSO3��ʵ��װ����ͼ��ʾ����ش�| �������� | �����뾧�������� | ���Ⱥ���������������� |
| 11.7g | 22.7g | 18.6g |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ��Ķ�����
 ʾ��λ�ڸ�������Ķ�������ģ��û�����Ļ�ѧʽ��
ʾ��λ�ڸ�������Ķ�������ģ��û�����Ļ�ѧʽ��
| Ԫ�� | Mn | Fe | |
| ���� ��/kJ?mol-1 |
��1 | 717 | 759 |
| ��2 | 1509 | 1561 | |
| ��3 | 3248 | 2957 |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
| a |
| 10 |
| 1.45 |
| a |
| 1.45 |
| a |
| 1.40 |
| a |
| 1.40 |
| a |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
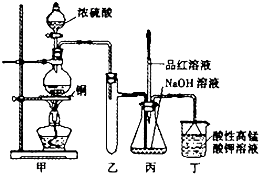 ʵ������Ũ������ͭ�ķ�Ӧ��ȡ����NaHSO3��ʵ��װ����ͼ��ʾ��
ʵ������Ũ������ͭ�ķ�Ӧ��ȡ����NaHSO3��ʵ��װ����ͼ��ʾ��| �������� | �����뾧�������� | ���Ⱥ���������������� |
| 11.7g | 22.7g | 18.6g |
�鿴�𰸺ͽ���>>
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com