
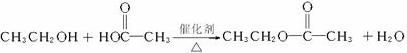
��֪�������ݣ�
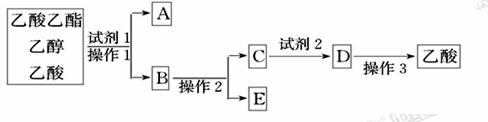
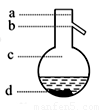
��ͼΪʵ������ȡ����������װ��ͼ��
(1)������̼������Һ�Ϸ��ռ����϶�Һ��ʱ��ֹͣ���ȣ�ȡ��С�Թ�B����������á���ǰ���ʵ������_________(��ѡ��)��
A.�ϲ�Һ��䱡 B.�²�Һ���ɫ��dz���Ϊ��ɫ
C.��������� D.�й���ζ
(2)Ϊ���������������Ҵ�������Ļ����ɰ����в�����з��룺
���Լ�1���ѡ��___________��
�ڲ���1��_____�����õ���Ҫ����������________��
���Լ�2���ѡ��___________��
�ܲ���2��___________��
�ݲ���3���¶ȼ�ˮ�����λ��ӦΪ����ͼ��______(��a��b��c��d)��ʾ���ڸò����У���������ƿ���¶ȼ��⣬����Ҫ�IJ���������_______��_______��_______��______���ռ�����������¶���___________��
(1)A��B��C��D
(2)�ٱ���̼������Һ �ڷ�Һ ��Һ©��
��ϡ���� ������
��b �ƾ��� ������ ţ�ǹ� ��ƿ �Ը���118 ��
����:��1����ȡ���������ķ�Ӧ�ǿ��淴Ӧ������������������˺������������⣬����������Ҵ���������Ժͱ���̼������Һ��Ӧ�ų�������̼���壬�Ҵ������ܽ��ڱ���̼������Һ�������������ܶ�С��ˮ�ģ��������ϲ㡣���A��B��C��D����ȷ��
��2����Ϊ����̼������Һ�����������ᣬ�ܽ��Ҵ���ͬʱ��������������ˮ�е��ܽ�ȣ����Կ���ͨ������̼������Һ��Һ����Һ����Ҫ�����Ƿ�Һ©����B����Ҫ���Ҵ���̼���ƺ������ƵĻ��Һ��ͨ������õ��Ҵ���Ҫ�õ����ᣬ��Ҫ�������������������ᣬ�����ͨ������õ����ᡣ����ʱ��Ҫ�����������¶ȣ������¶ȼƵ�ˮ����Ӧ����������ƿ֧�ܵij��ڴ�����b������Ϊ����ķе���118�棬�����¶�Ӧ�Ը���118�档



| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ��Ķ�����
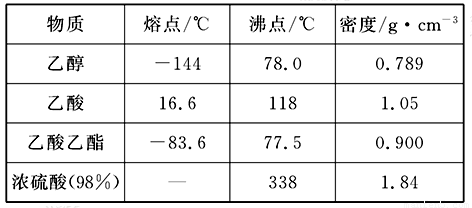
���� | �۵㣨 �棩 | �е㣨 �棩 | �ܶ�(g��cm-3) |
�Ҵ� | -117.0 | 78.0 | 0.79 |
���� | 16.6 | 117.9 | 1.05 |
�������� | -83.6 | 77.5 | 0.90 |
Ũ���ᣨ98���� | ���� | 338.0 | 1.84 |
ѧ����ʵ������ȡ������������Ҫ�������£�
����30 mL�Ĵ��Թ�A�а������1��4��4�ı�������Ũ���ᡢ�Ҵ�������Ļ����Һ��
�ڰ���ͼ���Ӻ�װ�ã�װ�����������ã�����С����ȵؼ���װ�л����Һ�Ĵ��Թ�5��10 min��
�۴��Թ�B�ռ���һ���������ֹͣ���ȣ������Թ�B��������Ȼ���ô��ֲ㡣
�ܷ�������������㣬ϴ�ӡ����
ͼ1 ͼ2
�������ĿҪ������������⣺
��1�����Ƹû����Һ����Ҫ��������Ϊ��___________________________________________��
д����ȡ���������Ļ�ѧ����ʽ��________________________________________________��
��2������ʵ���б���̼������Һ�������ǣ�����ĸ����_______________��
A.�к�������Ҵ�
B.�к����Ტ���ղ����Ҵ�
C.���������ڱ���̼������Һ�е��ܽ�ȱ���ˮ�и�С�������ڷֲ�����
D.�����������ɣ���������
��3�����������ҪС����ȼ��Ȳ���������Ҫ�����ǣ�_______________________________��
��4��ָ����������۲쵽������_____________________________������������������һ���ñ���ʳ��ˮ�ͱ����Ȼ�����Һϴ�ӣ���ͨ��ϴ�ӳ�ȥ�������ƣ�_____________ �����ʣ�Ϊ�˸�������������ѡ�õĸ����Ϊ������ĸ����_____________��
A.P2O5 B.��ˮNa2SO4 C.��ʯ D.NaOH����
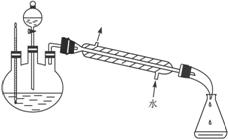
��5��ij��ѧ����С���������ͼ2��ʾ����ȡ����������װ�ã�ͼ�е�����̨�����С�����װ������ȥ������ͼ1װ����ȣ�ͼ2װ�õ���Ҫ�ŵ��У�_____________________��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ��2011-2012ѧ�����ɹŰ���һ�и߶���ѧ��4���¿���ѧ�Ծ����������� ���ͣ�ʵ����
��֪�������ݣ�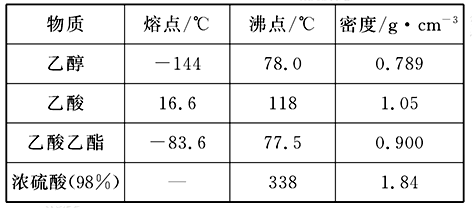
��ͼΪʵ������ȡ����������װ��ͼ��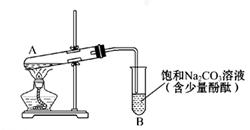
(1)������̼������Һ�Ϸ��ռ����϶�Һ��ʱ��ֹͣ���ȣ�ȡ��С�Թ�B����������á���ǰ���ʵ������_________(��ѡ��)��
| A���ϲ�Һ��䱡 | B���²�Һ���ɫ��dz���Ϊ��ɫ |
| C����������� | D���й���ζ |

�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ��2012���˽̰���л�ѧ����3.3 ���������ֳ������л�����ϰ���������棩 ���ͣ�ʵ����
��֪�������ݣ�
|
���� |
�۵�(��) |
�е�(��) |
�ܶ�(g/cm3) |
|
�Ҵ� |
��117.3 |
78.5 |
0.789 |
|
���� |
16.6 |
117.9 |
1.05 |
|
�������� |
��83.6 |
77.5 |
0.90 |
|
Ũ����(98%) |
�� |
338.0 |
1.84 |
ѧ����ʵ������ȡ������������Ҫ�������£�
����30 mL�Ĵ��Թ�A�а������1��4��4�ı�������Ũ���ᡢ�Ҵ�������Ļ����Һ��
�ڰ���ͼ���Ӻ�װ��(װ������������)����С����ȵؼ���װ�л����Һ�Ĵ��Թ�5��10 min��
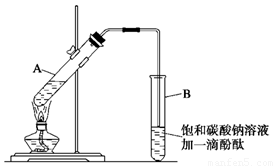
�۴��Թ�B�ռ���һ�����IJ����ֹͣ���ȣ���ȥ�Թ�B��������Ȼ���ô��ֲ㣻
�ܷ�������������㣬ϴ�ӡ����
�������ĿҪ��ش��������⣺
(1)���Ƹû����Һ����Ҫ��������Ϊ____________________________________
д����ȡ���������Ļ�ѧ����ʽ�� _____________________________________
(2)����ʵ���б���̼������Һ��������(����ĸ)_______________________________��
A���к�������Ҵ�
B���к����Ტ���ղ����Ҵ�
C�����������ڱ���̼������Һ�е��ܽ�ȱ���ˮ�и�С�������ڷֲ�����
D�������������ɣ���������
(3)���������ҪС����ȼ��Ȳ���������Ҫ������____________________________________
(4)ָ����������۲쵽������ _______________________________________________
���������������Ϊ�˸�������������ѡ�õĸ����Ϊ(����ĸ)__________��
A��P2O5 B����ˮNa2SO4 C����ʯ�� D��NaOH����
(5)ij��ѧ����С�����������ͼ��ʾ����ȡ����������װ��(ͼ�е�����̨�����С�����װ������ȥ)������ͼװ����ȣ���װ�õ���Ҫ�ŵ��У� __________________________________
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ��2013�����ɹŰ��и߶���ѧ��4���¿���ѧ�Ծ��������棩 ���ͣ�ʵ����
��֪�������ݣ�
��ͼΪʵ������ȡ����������װ��ͼ��

(1)������̼������Һ�Ϸ��ռ����϶�Һ��ʱ��ֹͣ���ȣ�ȡ��С�Թ�B����������á���ǰ���ʵ������_________(��ѡ��)��
A.�ϲ�Һ��䱡 B.�²�Һ���ɫ��dz���Ϊ��ɫ
C.��������� D.�й���ζ
(2)Ϊ���������������Ҵ�������Ļ����ɰ����в�����з��룺

���Լ�1���ѡ��___________��
�ڲ���1��_____�����õ���Ҫ����������________��
���Լ�2���ѡ��___________��
�ܲ���2��___________��
�ݲ���3���¶ȼ�ˮ�����λ��ӦΪ����ͼ��______(��a��b��c��d)��ʾ���ڸò����У���������ƿ���¶ȼ��⣬����Ҫ�IJ���������_______��_______��_______��______���ռ�����������¶���___________��
�鿴�𰸺ͽ���>>
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com