
NA��ʾ�����ӵ�������ֵ������˵����ȷ���м���
���ڳ��³�ѹ�£�1mol��������ԭ����Ϊ2NA
��0.5molO2��0.5molO3�Ļ���������е���ԭ�Ӹ���Ϊ2.5NA
�۳��³�ѹ�£�18 g H2O�к��е�ԭ������Ϊ3NA
�ܳ��³�ѹ�£�2.24 LCO��CO2��������к��е�̼ԭ����ĿΪ0.1NA
�ݺ�NA��Na����Na2O�ܽ���1 Lˮ�У�Na�������ʵ���Ũ��Ϊ1 mol��L��1
��1.00 mol NaCl�У�����Na����������������Ϊ8��6.02��1023
��1 mol��L��1 NaCl��Һ����NA��Na��
�ೣ�³�ѹ�£�92 g��NO2��N2O4������庬�е�ԭ����Ϊ6NA
��13.5g Al���������ᷴӦ����H2�ķ�����ΪNA
���״���£�2.24LCH4����������ΪNA
A��3 B��4  C��5 D��6
C��5 D��6
 �Ķ��쳵ϵ�д�
�Ķ��쳵ϵ�д�
| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ��2016-2017ѧ�����ʡ�߶������л�ѧ���������棩 ���ͣ�ʵ����
������к͵ζ�������֪ijNaOH�����к���NaCl���ʣ�Ϊ�ⶨ������NaOH�������������������²���ʵ�飺
�ٳ���1.00g��Ʒ����ˮ�����250 mL��Һ��
��ȷ��ȡ25.00mL������Һ����ƿ�У�
�� �μӼ��η�̪��Һ��
����0.10mol��L�ı�����ζ����Σ�ÿ����������������¼���£�
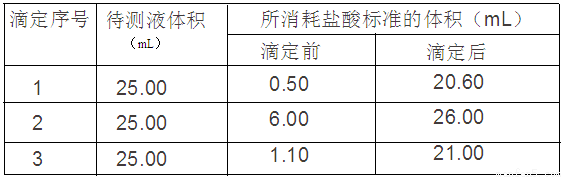
��1����___________�ζ��ܣ����ʽ����ʽ����ʢװ0.10mol/L�������Һ��
��2��������NaOH����������Ϊ______________________��
��3������������������ⶨ���ƫ�ߵ���___________��
a���ζ�ǰ������ˮ��ϴ��ƿ
b��������ƿʱ������ƿ����Һ����
c�����ڵζ������в�����������Һ������ƿ��
d����ʽ�ζ��ܵ����յ�ԣ����Ӷ���
e����ʽ�ζ���������ˮϴ��δ�ñ�Һ��ϴ
��������ԭ�ζ���ȡ������Һ������ƿ�У���������ϡ���ᣬ��Ũ��Ϊ0.1mol��L��1�ĸ��������Һ�ζ��������ķ�ӦΪ��2KMnO4+5H2C2O4+3H2SO4=K2SO4+10CO2��+2MnSO4+8H2O��
��4���ζ�ʱ��KMnO4��ҺӦװ����ʽ�ζ����У��ζ��յ�ʱ�ζ�������________________��
�����ζ��D�D�ζ����ͱ��ζ����������ȵζ�����ָʾ��������������ܡ�
��5���ο��±��е����ݣ�����AgNO3�ζ�NaSCN��Һ����ѡ�õ�ָʾ���� ����ѡ����ĸ����
������ | AgCl | AgBr | AgCN | Ag2CrO4 | AgSCN |
��ɫ | �� | dz�� | �� | ש�� | �� |
Ksp | 1.77��10��10 | 5.35��10��13 | 1.21��10��16 | 1.12��10��12 | 1.0��10��12 |
A��NaCl B��NaBr C��NaCN D��Na2CrO4
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ��2016-2017ѧ�������ĵ������ѧ�߶��������Ļ�ѧ���������棩 ���ͣ�ѡ����
��ѧ������������أ�����˵���в���ȷ���ǣ� ��
A. Al2O3 �۵�ߣ�������������������
B. ˮ�����߲˵��Ǽ���ʳ��
C. �ߴ��ȵĹ赥�ʿ������������ά
D. ��ɫ��ѧ�ĺ����Ǵ�Դͷ�϶ž���Ⱦ�IJ���
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ��2016-2017ѧ�������ĵ������ѧ�߶�����������ѧ���������棩 ���ͣ�ѡ����
��Ӧ X��g���� Y��g�� 2Z��g��������Ӧ���ȣ�,��һ�������£���Ӧ��Y��ת�����뷴Ӧʱ�䣨t���Ĺ�ϵ��ͼ��ʾ����ʹ����a��Ϊ����b���ɲ�ȡ�Ĵ�ʩ�ǣ��� ��
2Z��g��������Ӧ���ȣ�,��һ�������£���Ӧ��Y��ת�����뷴Ӧʱ�䣨t���Ĺ�ϵ��ͼ��ʾ����ʹ����a��Ϊ����b���ɲ�ȡ�Ĵ�ʩ�ǣ��� ��
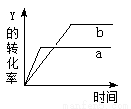
A.������� B.�����¶� C.����ѹǿ D.����Y��Ũ��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ��2016-2017ѧ�������ĵ������ѧ�߶�����������ѧ���������棩 ���ͣ�ѡ����
����ͬ�¶�ʱ��100mL0.01mol•L-1�Ĵ�����Һ��10mL 0.1mol•L-1�Ĵ�����Һ��Ƚϣ�������ֵ�У�ǰ�ߴ��ں��ߵ��ǣ� ��
A. H+�����ʵ��� B. ����ĵ��볣��
C. �к�ʱ����NaOH���� D. CH3COOH�����ʵ���
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ��2017��ӱ�ʡ�����ϵ������¿���ѧ�Ծ��������棩 ���ͣ�ѡ����
����˵������ȷ����
A���ȵĴ�����Һ����Ĵ�����Һȥ����Ч����
B��ʢ��Na2CO3��Һ���Լ�ƿ����������
C����AlCl3��Һ���ȡ����ɡ����գ��ɵõ�����Al2O3
D�����ڳ�ʪ�Ļ���������������ˮ����
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ��2017��ӱ�ʡ�����ϵ������¿���ѧ�Ծ��������棩 ���ͣ�ѡ����
��ʵ�ϣ�������������һ������������Na2O2��Ӧ���ҷ�Ӧ���й��ɣ���Na2O2+SO2=Na2SO4��2Na2O2+2SO3=2Na2SO4+O2���ݴ�����Ϊ���з�Ӧ����ʽ�в���ȷ����
A��2Na2O2+2N2O5=4NaNO3+O2�� B��Na2O2+2NO2=2NaNO2+O2
C��Na2O2+N2O4=2NaNO3 D��2 Na2O2+2Mn2O7=4NaMnO4+O2��
Na2O2+2Mn2O7=4NaMnO4+O2��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ��2017��ӱ�ʡ�����ϵ������¿���ѧ�Ծ��������棩 ���ͣ�ѡ����
��ͼΪN2(g)��O2(g)��Ӧ����NO(g)�����е������仯������˵����ȷ����
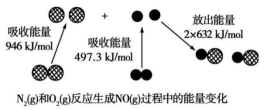
A��ͨ������£�NO��N2�ȶ�
B��ͨ������£�N2(g)��O2(g)�����ֱ�ӷ�Ӧ����NO
C��1 mol N2(g)��1 mol O2(g)�����������2 mol NO(g)�������
D��1 mol N2(g)��1 mol O2(g)��Ӧ�ų�������Ϊ179.3 kJ
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ��2016-2017ѧ������ʡ�߶������л�ѧ�Ծ��������棩 ���ͣ�ѡ����
һ�������µ�2L�ܱ����������з�Ӧ2X��g��+Y��g��  2Z��g��������ʼʱX��Y��Z���ʵ����ֱ�Ϊn1��n2��n3������Ϊ�㣩����ƽ��ʱ��X��Y��ZŨ�ȷֱ�Ϊ0.2mol/L��0.1 mol /L��0.08 mol/L���������жϺ������ǣ� ��
2Z��g��������ʼʱX��Y��Z���ʵ����ֱ�Ϊn1��n2��n3������Ϊ�㣩����ƽ��ʱ��X��Y��ZŨ�ȷֱ�Ϊ0.2mol/L��0.1 mol /L��0.08 mol/L���������жϺ������ǣ� ��
A�� X��Y��ת���ʲ�һ�����
B�� n1��n2=2:1
C�� ƽ��ʱ��Y��Z����������֮��Ϊ2:1
D�� n1��ȡֵ��ΧΪ0<n1<0.28
�鿴�𰸺ͽ���>>
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com