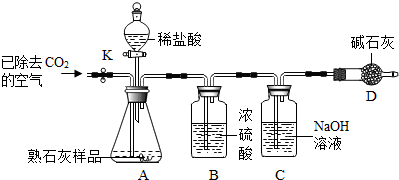
ͬѧ�ǶԿα��ϡ���Խ�ǿ�Ľ���һ����Խ�λ�������Ľ��������ǵ�����Һ���û��������Լ���֧��ȼ�յ�����һ���������������仰���������ɣ�����ʦ�İ����£��ý�������̽���������⣺
[�������]1���������ܷ��û�������ͭ��Һ�е�ͭ��2���������ܷ����������壨�磺CO
2����ȼ�գ�
[��������]����Na���ʻ��ã������£���������������Ӧ���ɰ�ɫ���������ƣ�����������ˮ�����������ƣ������ƻ�������ˮ���ҷ�Ӧ�ų�������
[ʵ��̽��]ʵ��һ������ʵ�����У������Ʊ�����ú���У��������Ӵ�ú����ȡ��һС������ƣ�����ֽ���ɱ���ú�ͺ�Ͷ������ͭ��Һ�У��۲쵽��Һ�г�����ɫ��������û�к�ɫ������������
�۰��ղ���ڽ�������Ͷ��ˮ�У�����Ӧ������μӷ�̪��Һ��������Һ��죮
ʵ�������ȼ�յ���Ѹ������ʢ��CO
2�ļ���ƿ�У��۲쵽����CO
2�м���ȼ�գ���Ӧ����ȴ��ƿ�ڸ��ź�ɫ��������ճ���Ű�ɫ���ʣ�
[��������]��1�������Ʊ�����ú���е�ԭ��
������������Ӧ����
������������Ӧ����
��
��2��������������д���ƿ�Ͷ��ˮ���������ķ�Ӧ����ʽ��
2Na+2H2O�T2NaOH+H2��
2Na+2H2O�T2NaOH+H2��
����Ͷ������ͭ��Һ���ܵķ�Ӧ����ʽ��
2Na+2H2O+CuSO4=H2��+Cu��OH��2��+Na2SO4
2Na+2H2O+CuSO4=H2��+Cu��OH��2��+Na2SO4
��������ͭ���ȷֽ������ͭ��ˮ������24.0gʣ�����������¼��ȣ����ֹ�������������2.4g���������ٸı䣬д���˹����п��ܷ�����Ӧ�Ļ�ѧ����ʽ��
��
��3��ʵ��������ɵĺ�ɫ������
C
C
������ɫ���ʿ�����ʲô�أ�
��ͬѧ��Χ�ư�ɫ���ʿ�����ʲô�������²��룺����Ϊ��ɫ������Na
2O��������Na
2CO
3��������Na
2O��Na
2CO
3�Ļ�������Ϊ��NaOH��
����Ϊ��Ϊ���IJ����Ǵ���ģ�������
��Ӧ����û����Ԫ��
��Ӧ����û����Ԫ��
��
�ڶ�ɫ������ʲô����ʵ����̽��������������£�
����1��ȡ����Ʒ�������Թ������������ˮ�������ˣ�����Һ�м�����ɫ��̪��Һ���۲쵽��Һ��ɺ�ɫ�����ۣ���ɫ������һ����Na
2O��
����2��ȡ����Ʒ�������Թ���μ�����ϡ���ᣬ��Һ��������ð�������ۣ���ɫ������һ����
̼����
̼����
��
����3��ȡ����Ʒ�������Թ������������ˮ�������ˣ�����Һ�м��������
B
B
��Һ����ѡ�Լ���A��ʯ��ˮB���Ȼ�����Һ C������ͭ��Һ�������ְ�ɫ������Ȼ����ˣ�������Һ��μ���ɫ��̪��Һ��������������֣���
��
��
���ڼס��ҡ�����ѡ���IJ��������
�������뷴˼�������Ϊʵ�鷽��1�ó��Ľ��۲���ȷ����������
̼������ҺҲ�Ǽ��Ե�
̼������ҺҲ�Ǽ��Ե�
��
ʵ�����еĽ������Ż�ʱ����������
�û�ɳ����
�û�ɳ����
��
[��չӦ��]С��ͬѧΪ�ⶨʵ����������õı�ʾ��������Ϊ32%������ ��ͼ1����ʵ����������������С�������ͬѧ��pH�ⶨ�����ʵ��װ�ã�ͼ2����ʵ��ʱ�����ձ��м���20g 40%������������Һ������μ��˸����ᣬ�ⶨ�Ǵ�ӡ������������������ձ�����ҺpH�Ĺ�ϵͼ��ͼ3����

�ٷ�����Ӧ�Ļ�ѧ����ʽΪ��
HCl+NaOH=NaCl+H2O
HCl+NaOH=NaCl+H2O
_��
�����Դ˴βⶨ�Ľ�����ɼ�����������ʵ������������
29.2%
29.2%
��
���������ļ��������ǩ��ʾ������������һ�µĿ���ԭ��
Ũ������лӷ��ԣ����ڷ�������������С
Ũ������лӷ��ԣ����ڷ�������������С
��



 ȫ�ܲ����ĩС״Ԫϵ�д�
ȫ�ܲ����ĩС״Ԫϵ�д�