
ij����С��Ϊ�ⶨ����ʯ��ʯ�к�̼��Ƶ�����������ȡ����һЩ��ʯ��Ʒ����ȡϡ����200g��ƽ���ֳ�4�ݣ����ķݲ�ͬ��������Ʒ���뵽�ķ�ϡ�����У�����ʵ�飬������£�
|
ʵ�� |
1 |
2 |
3 |
4 |
|
������Ʒ������/g |
5 |
10 |
15 |
20 |
|
����CO2������/g |
1��54 |
3��08 |
4��4 |
m |
��1���ϱ���m����ֵ��__________________��
��2���Լ�������ʯ��ʯ����̼��Ƶ�����������
��3�������η�Ӧ��������Һ���������������Ƕ��٣�
��1��4��4�� ��2��70% ��3��20%
��������
����������Աȱ������ݿ�֪��ʵ��1��ʵ��2�е�����û����ȫ��Ӧ�������е�̼��Ʒ�Ӧ��ȫ��ʵ��3��ʵ��4�е����ᷴӦ��ȫ�����е�̼���û�з�Ӧ��ȫ�����Ե�4��ʵ���е�m��Ȼ��4��4g����5gʯ��ʯ��������̼��Ƶ�����Ϊx����
CaCO3+2HCl=CaCl2+H2O+CO2��
100 44
x 1��54g
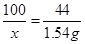
x=3��5g
ʯ��ʯ����̼��Ƶ���������=3��5g/5g��100%=70%��������η�Ӧ�����Ȼ��Ƶ�����Ϊy���μӷ�Ӧ��̼��Ƶ�����Ϊz����
CaCO3+2HCl=CaCl2+H2O+CO2��
100 111 44
z y 4��4g
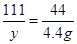
y=11��1g
100/z = 44/4��4g z=10g
�����η�Ӧ��������Һ��������������=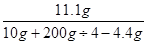 ��100%=20%��
��100%=20%��
���㣺�����ʵķ�Ӧ��������������ļ��㣬���ʵ�����������



| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
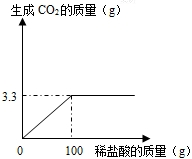 ij����С��Ϊ�ⶨ����ʯ��ʯ�к�̼��Ƶ�����������ȡ����һЩ��ʯ��ȷ��ȡ��Ʒ10g�����ձ��У����ʼȲ�����ˮ��Ҳ����ϡ���ᷴӦ��Ҳ���ֽ⣩�������м���������ϡ���ᣬ����ʵ���õ����ݻ�����ͼ��
ij����С��Ϊ�ⶨ����ʯ��ʯ�к�̼��Ƶ�����������ȡ����һЩ��ʯ��ȷ��ȡ��Ʒ10g�����ձ��У����ʼȲ�����ˮ��Ҳ����ϡ���ᷴӦ��Ҳ���ֽ⣩�������м���������ϡ���ᣬ����ʵ���õ����ݻ�����ͼ���鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
| ʵ�� | 1 | 2 | 3 | 4 |
| ������Ʒ������/g | 5 | 10 | 15 | 20 |
| ����CO2������/g | 1.54 | 3.08 | 4.4 | m |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
| c-b |
| a |
| c-b |
| a |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
| ʵ�� | 1 | 2 | 3 | 4 |
| ������Ʒ������/g | 5 | 10 | 15 | 20 |
| ����CO2������/g | 1.54 | 3.08 | 4.4 | m |
�鿴�𰸺ͽ���>>
����ѧУ��ѡ - ��ϰ���б� - �����б�
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com