
����˵����ȷ���� ( )
A. �кͷ�Ӧ���κ�ˮ���ɣ�������κ�ˮ���ɵķ�Ӧһ�����кͷ�Ӧ
B. �������к�����Ԫ�أ�������Ԫ�صĻ����ﲻһ����������
C. ���ж�������Ԫ�أ����Ժ�����Ԫ�صĻ�����һ������
D. �û���Ӧһ���е������ɣ��е������ɵ�һ�����û���Ӧ
B ��������A����ͼ������κ�ˮ�ķ�Ӧ���кͷ�Ӧ��������������ﷴӦҲ�������κ�ˮ���������кͷ�Ӧ������B��������Ԫ�����������һ������Ԫ�صĻ�������������������������Ԫ�أ��������������ȷ��C������ˮ������������ֻ�������ӵĻ�������ᣬ��������������Ԫ�أ��������ᣬ����D��һ�ֵ��ʺ�һ�ֻ�����������һ�ֵ��ʺ���һ�ֻ�����ķ�Ӧ���û���Ӧ�����ˮ�����������������е������ɣ������û�... �ƸԹھ��ο���ϵ�д�
�ƸԹھ��ο���ϵ�д�
| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ���ӱ�ʡ�Ϲ���2018����꼶��ѧ�ڵڶ����¿���ѧ�Ծ� ���ͣ������
ij��ȤС��Ҫ�ⶨijʯ��ʯ��Ʒ�����ʵ��������������ǽ�������ͼʵ�顣(���ʲ�������Ӧ)
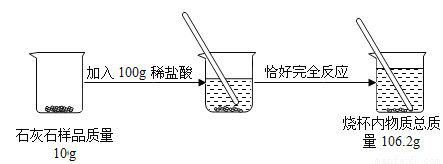
����㣺
(1)����CO2���������Ϊ________��
(2)��Ʒ�����ʵ���������Ϊ________��
3.74g 15% ��������������������������غ㶨�ɿ�֪����Ӧ��ȷ�Ӧǰ���ٵ�������Ϊ��Ӧ�ų�������̼����������� ���ݻ�ѧ����ʽ�ɶ�����̼���������Լ����ʯ��ʯ��̼��Ƶ��������Ӷ����������Ʒ�����ʵ����������� ���������������غ㶨�ɣ�����CO2���������Ϊ��10g+100g-106.26g=3.74g�� �������������� ��ʯ��ʯ��̼��Ƶ�����Ϊx 100 44 ...�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ʡ��������Ƭ2018����꼶��ѧ�ڵڶ����¿���ѧ�Ծ� ���ͣ��ۺ���
ijЩʳƷ�İ�װ���ڣ�����װ�а�ɫ����״�����Сֽ��������д�š����������Ҫ�ɷ�Ϊ��ʯ�ҡ���ijͬѧ��һ�����ÿ����еĸ����������̽����
����ȡ���־��ø������������ˮ�У���ֽ��衢����,�õ���ɫ�������Һ��Ȼ�����Һ������ԺͰ�ɫ������������̽����
��ʵ��̽��һ��̽����Һ�������
��ͬѧ�ⶨ����Һ������ԣ�����Ϊ����Һ��____________�ԡ�
��ʵ��̽������̽�����˺��ɫ��������
��ͬѧ��Ϊ��ɫ�����п��ܺ��е�������CaO��Ca(OH)2��CaCO3��
�������й����ϵ�֪��CaO��һ�ְ�ɫ���壬�׳���ʯ�ң�����ˮ��Ӧ�����������ƣ����ų�������������������ˮ��
�������жϣ�����Ϊ�ù��˺�õ��İ�ɫ������һ�������е�������_______��
��ʵ������������Ǹ�ͬѧ��ƵĽ�һ��̽����ɫ������ɵ�ʵ�鷽�����������һ�����ʵ�鱨�档
ʵ����� | ʵ������ | ʵ����� |
��ȡ������ɫ���壬��������ˮ����ֽ��衢���ã� �����ϲ���Һ�еμ���ɫ��̪��Һ | �ϲ���Һ��____ɫ | ��ɫ������һ�������������� |
��ȡ������ɫ��������Թ��У��μ�ϡ���� | _________ | ��ɫ������һ������̼��� |
���ܽᷴ˼��
ֻͨ������ʵ�飬������֤����������ڿ����еĸ�������Ƿ���CaO���������һ��֤�����������к��н϶���CaO�ļ���ʵ�飺____���û�ѧ����ʽ��ʾ���еķ�Ӧ_____��
��ʵ��̽������̽���������Ʒ��̼��Ƶ���������
ȡ�����������Ʒ10g������ͼ��װ���У���ַ�Ӧ�������Ͳ�е�ˮ�����Ϊ220mL,(��״���¶�����̼���ܶ���2g/L),��˸������̼��Ƶ���������Ϊ___________(ͨ��������գ�
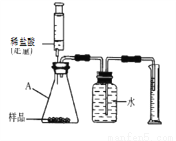
���ܽᷴ˼����ʵ��̽��������ʵ����ƵIJ�����֮��_________
���ԡ� �����ƻ�CaO �� ��������� ֤�����������к��н϶���CaO�ļ���ʵ�飺ȡ������Ʒ���Թ��У���ˮ�ܽ⣬�����Թ���ڣ��¶����� CaO+H2O==Ca(OH)2 10% ���ɵĶ�����̼����ˮ����ˮ������Ӧ��ʹ������̼���������С ����������ʵ��̽��һ��������õĸ�����к��������ƣ������������ƣ����������ƺ��������ƣ���������ˮ�У���ֽ��衢���˺����Һ�к����������ƣ���Һ�Լ���...�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ��ɽ��ʡ������2018����꼶��ѧ�ڵڶ����������Ի�ѧ�Ծ� ���ͣ������
С����Ҫ����80g��������Ϊ10%������������Һ��������Ҷ����ǩ��
��1�����㣺��Ҫ�������ƹ���______g��ˮ_______mL��ˮ���ܶ���1.0g/cm3�ƣ�
��2������20%������������Һ���ƣ���Ҫ��Һ������Ϊ__g
8 g�� 72 ml 40g ����������1����Ҫ�������ƹ��壺80g��10%=8g��ˮ��80g -8g=72g����72mL��ˮ���ܶ���1.0g/cm3�ƣ�����2������20%������������Һ���ƣ���Ҫ��Һ������Ϊx��20%x=80g��10% X=40g�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ��ɽ��ʡ������2018����꼶��ѧ�ڵڶ����������Ի�ѧ�Ծ� ���ͣ������
����H��O��C��C1��Na����Ԫ�أ���ѡ�����е�һ�ֻ���Ԫ����д��������Ҫ��Ļ�ѧʽ��ѧ����ʽ(��дһ��)��
(1)����θҺ�к��е��� ________��
(2) �����õ��� _________��
(3) ������ˮ�õ�С�մ�__________��
(4)�ƾ��Ļ�ѧʽ__________________��
(5)���Է������кͷ�Ӧ _______________________
HCl NaCl NaHCO3 C2H5OH HCl+NaOH�TNaCl+H2O ��������������Ļ�ѧʽ��д�������۵�Ԫ�������д����ߣ��Ը��۵�д���ұߣ����ϼ۵ľ���ֵ����Լ�����û�ѧʽ���½ǵ����֣�����Ϊ1ʱ��д��(1)����θҺ�к��е��������ᣬ����Ϊ�Ȼ��⣬��ѧʽΪ��HCl��(2) �����õ�����ʳ�Σ���ѧʽΪ��NaCl��(3) ������ˮ�õ�С�մ���̼�����ƣ���ѧʽΪ��NaHCO3��(4...�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ��ɽ��ʡ������2018����꼶��ѧ�ڵڶ����������Ի�ѧ�Ծ� ���ͣ���ѡ��
���й����������ӵĽṹʾ��ͼ��˵������ȷ���ǣ� ��

�� �� �� ��
A���٢����ڲ�ͬ��Ԫ�� B���ڢ۵Ļ�ѧ��������
C�����������ӣ����ӷ���ΪMg2+ D���ٱ�ʾ����ϡ������Ԫ��
C �������� ���������A��Ԫ���Ǿ�����ͬ���������˵��������ͬһ��ԭ�ӵ��ܳƣ��ʾ���Ԫ��������������������Ԣ٢�����ͬ��Ԫ�أ�����B����������������ԭ�ӵĻ�ѧ���ʣ���������������ȣ���ѧ�������ƣ��ں͢۵���������������ȣ��ʻ�ѧ���ʲ����ƣ�����C���ܵ�������Ϊ12�����������Ϊ10��������2����������ɣ��������ӣ����ӷ���ΪMg2+����ȷ��D���ٵ�������Ϊ17�����������Ϊ...�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ���Ĵ�ʡ������2017����꼶��ѧ����ĩ���������ۺϻ�ѧ�Ծ� ���ͣ������
�����ǵ���Ҫ�ɷ���̼��ƣ�����Ϊ���ʣ�(���ʲ�����ˮ�Ҳ��μӷ�Ӧ)Ϊ�˲ⶨ������̼��Ƶ�����������С����С��ͬѧ����������ʵ�飬���ʵ����������ͼ��ʾ��
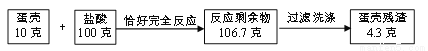
(1)С�����ݡ����Dz�������Ϊ4.3�ˡ�����õ�����̼��Ƶ���������Ϊ_________��
(2)С�����ݡ���Ӧ�����м��ٵ�������(10��+100��-106.7��=3.3��)Ҳ����˵�����̼��Ƶ���������������д��С���ļ�����̺ͽ��__________________��
(3)С����С����������ʵ��ó��ü�������̼��Ƶ�����������С������Ǻϣ������С����ʼ���ý����������ԭ����__________________________��
57% 75% С���������õĵ��Dz���δ���� ��������(1)С�����ݡ����Dz�������Ϊ4.3�ˡ�����õ�����̼��Ƶ���������Ϊ��10g-4.3g����10g��100%=57%��(2)���������غ㶨�ɣ��ڻ�ѧ��Ӧ�У��μӷ�Ӧǰ�����ʵ������ܺ͵��ڷ�Ӧ�����ɸ����ʵ������ܺͣ��������ɶ�����̼������=�μӷ�Ӧǰ�����ʵ������ܺͷ�Ӧ��ʣ������ʵ������ܺͣ����ɶ�����̼������Ϊ��10g+100g-106....�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ��������2018��ָ�굽У�������Ի�ѧ�Ծ� ���ͣ������
(6��) ijͬѧ����֤�������ǵ���Ҫ�ɷ���̼���Σ����ռ�������������塱��ʵ�顣��������·�������ʵ�飺(�Ƶõ�CO2�г�����HCl��ˮ����)
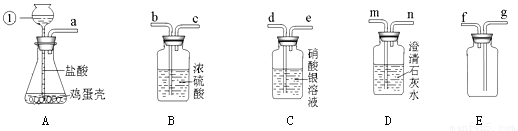
д���������������ƣ��� ��
����A��������� ��
����������ѧ��֪ʶ������ΪAװ�ú� װ������������������ʱ������˵�������ǵ���Ҫ�ɷ���̼���Σ�д���������з�����Ӧ�Ļ�ѧ����ʽ_______________��װ
��ͬѧ��Ҫ�ռ�һƿ��������ĸ����壬��������װ�õ�˳���ǣ�����д���ӿ���ĸ��a�� �����У�д��Cװ�õ������ǣ� ��
��1�� ����©�� ��2�� �����ݲ����������ܽ� ��3�� D Ca(OH)2+ CO2 = CaCO3��+ H2O ��4�� a��d��e��b��c��g��(f)��f���п��ޣ� ����HCl �������� �����������1���������������ƣ��ٳ���©������2����ӦʱA�е�����Ϊ�����ݲ����������ܽ⣻��3������������ѧ��֪ʶ������ΪAװ�ú�Dװ������������������ʱ������˵�������ǵ���Ҫ�ɷ���̼���Σ�����...�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ���Ĵ�ʡüɽ��2018����꼶�п�ģ��(һ)��ѧ�Ծ� ���ͣ�������
С�����ñ��ǣ���Ҫ�ɷ�Ϊ̼��ƣ���ȡ������̼���塣Ϊ�ⶨ������̼��Ƶĺ���������������ʵ�飺���ȳ�ȡ50g������Ʒ���ձ��У��ձ�����Ϊ100�ˡ�Ȼ��150g������ƽ����5�μ��루�������ʾ�������ˮҲ�������ᷴӦ�����ձ��С�ʵ������е����ݼ�¼���£�
ʵ����� | ��һ�� | �ڶ��� | ������ | ���Ĵ� | ����� |
�ձ�+ʣ���������� | 175.6g | X | 226.8g | 252.4g | 282.4g |
��1��X��_____________g��
��2��������Ʒ��̼��Ƶ���������__________��
��3�����Ĵ�ʵ���������Һ�����ʵ���������_________��
201.2 80% 31.2% ��������(1)�ڶ��μ���ϡ������ձ�+ʣ�����ʡ�����Ϊ��175.6g+30g-4.4g=201.2g��(2)��Ӧ�в����Ķ�����̼��������Ϊ50g+100g+150g-282.4g=17.6g����50g������Ʒ��̼��Ƶ�����Ϊx�� CaCO3+2HCl=CaCl2+H2O+CO2�� 100 44 x 17.6g x=40g�� ����...�鿴�𰸺ͽ���>>
����ѧУ��ѡ - ��ϰ���б� - �����б�
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com