
��һ��ʯ��ʯ��Ʒ�����к��е������Ƕ�������
(����������һ�ֲ�����ˮ���������ᷴӦ�����µĹ���)��ijͬѧ��ⶨ����Ʒ�Ĵ��ȣ���ȡ��2g������ʯ��ʯ��Ʒ����20gϡ�����4�μ��룬��ַ�Ӧ��ʣ�������������±����ʣ�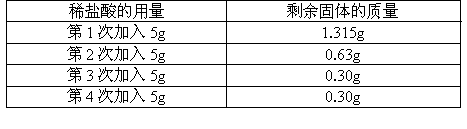
(1)2g
ʯ��ʯ��Ʒ�У������ʶ��ٿˣ�(2)
ʯ��ʯ��Ʒ�Ĵ����Ƕ��٣�(3)100kg
���ִ��ȵ�ʯ��ʯ������պɵõ��������ǧ�ˣ�(4)
�μӷ�Ӧ��ϡ������������������Ƕ��٣�
| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
| ʵ����� | ��1�� | ��2�� | ��3�� | ��4�� |
| ��������������/g | 50.00 | 50.00 | 50.00 | 50.00 |
| ʯ��ʯ��Ʒ������/g | 5.00 | 10.00 | 15.00 | 20.00 |
| ͨ�����㣬���ɵĶ�����̼������/g | 1.76 | 3.52 | 4.40 | 4.40 |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
| ||
| ||
| ��Ӧǰ | ��Ӧ�� | ||
| ʵ������ | �ձ���ϡ��������� | ʯ��ʯ��Ʒ������ | �ձ������л��������� |
| 160g | 12g | 167.6g | |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
| ʵ����� | ��1�� | ��2�� | ��3�� | ��4�� |
| ��������������/g | 50.00 | 50.00 | 50.00 | 50.00 |
| ʯ��ʯ��Ʒ������/g | 5.00 | 10.00 | 15.00 | 20.00 |
| ͨ�����㣬���ɵĶ�����̼������/g | 1.76 | 3.52 | 4.40 | 4.40 |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ������
| ʵ����� | ��1�� | ��2�� | ��3�� | ��4�� |
| ��������������/g | 50.00 | 50.00 | 50.00 | 50.00 |
| ʯ��ʯ��Ʒ������/g | 5.00 | 10.00 | 15.00 | 20.00 |
| ͨ�����㣬���ɵĶ�����̼������/g | 1.76 | 3.52 | 4.40 | 4.40 |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ��2004-2005ѧ�����ʡ��ɳ�г�����ѧ���꼶���ϣ��¿���ѧ�Ծ���12�·ݣ��������棩 ���ͣ������
| ʵ����� | ��1�� | ��2�� | ��3�� | ��4�� |
| ��������������/g | 50.00 | 50.00 | 50.00 | 50.00 |
| ʯ��ʯ��Ʒ������/g | 5.00 | 10.00 | 15.00 | 20.00 |
| ͨ�����㣬���ɵĶ�����̼������/g | 1.76 | 3.52 | 4.40 | 4.40 |
�鿴�𰸺ͽ���>>
����ѧУ��ѡ - ��ϰ���б� - �����б�
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com