
��13�֣�����A ~ F�dz��л�ѧ�е�����ʵ�飬�밴Ҫ����գ�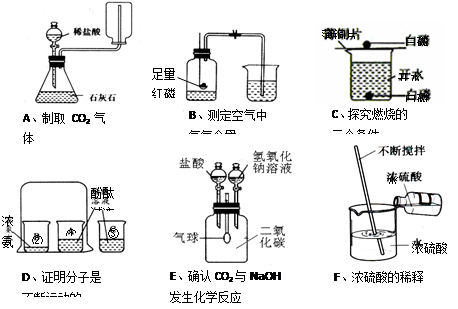
��1��Dʵ�����ձ��۵������� ��Fʵ���н����Ŀ���� ��
��2��Bʵ���к���ȼ�յĻ�ѧ����ʽΪ ��ʵ��˵�����������Լռ������ ��ʵ��ɹ��Ĺؼ��� ������ţ���
��װ�������Ժã���ʵ��ǰ�н�ֹˮ�У��ۺ�����������������ȴ���ٴ�ֹˮ�У���Ҫѡ�ý����ڵĿ�����
��3��Eʵ���ȫ���̹۲쵽��������
��������Ӧ��ѧ����ʽΪ ��
��4������ʵ�����ܴﵽʵ��Ŀ������ȷ���� ������ĸ����
���13��
��1�����ԱȻ�Ա�ʵ�飨1�֣���ɢ�ȣ�ʹ����������Ѹ����ɢ������ȣ�1�֣�
��2��4P + 5O2 ��ȼ 2P2O5��2�֣��� 1/5 ��1�֣����٢ڢۢܣ�ȫ��1�֣������֡���
��3���ٵ�������������Һ���������ͣ��ڵ�������������ݣ���������С���������2�㼴��1�֣�
CO2 + 2NaOH ="=" Na2CO3 + H2O��2�֣�
Na2CO3 + 2HCl ="=" 2NaCl + H2O + CO2����2�֣�
��4��B��D��E�����������������1�֣�ȫ��2�֣���������ѡ����Ϊ0�֡���
����



| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ�

�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�

�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�

�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�

| ||
| ||
| ||
| ||
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�

| ||
| ||
�鿴�𰸺ͽ���>>
����ѧУ��ѡ - ��ϰ���б� - �����б�
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com