ij����Ʒ����Ч�ɷ�Ϊ̼��ƣ������ɷֲ�����Ԫ�أ���˵���б���������Ϊ30%������ͬѧΪ�˼���ò�Ʒ��ȡ10g����Ʒ����100gϡ�����У�HCl�벹��Ʒ����Ч�ɷָպ���ȫ��Ӧ�������ɷֲ������ᷴӦ������Ӧ��ʣ������Һ����������ȷ�Ӧǰ������2.2g����������⣺
��1����Ӧ�Ļ�ѧ����ʽΪ______
��2�����ɶ�����̼������Ϊ______
��3������Ʒ��̼��Ƶ����������Ƕ��٣���д��������̣�
��4������Ʒ��ʵ�ʺ��Ƶ����������Ƕ��٣���д��������̣�
���𰸡�
������������Ӧ�Ĺ��̿�֪����Ӧ����Ψһ���������̼�ķų���ɷ�Ӧ��ʣ������Һ����������ȷ�Ӧǰ������2.2g������жϷų�������̼���������Ϊ2.2g�����÷�Ӧ�Ļ�ѧ����ʽ���ɶ�����̼��������ɸպ���ȫ��Ӧʱ�μӷ�Ӧ��̼��������ļ��㣻Ȼ����̼����и�Ԫ�ص������벹��Ʒ�����ȼ��㲹��Ʒ��ʵ�ʺ��Ƶ�����������
����⣺��1���������Ϣ��֪�÷�Ӧ�ķ���ʽΪCaCO
3+2HCl=CaCl
2+H
2O+CO
2����
��2��������Ӧ�Ĺ��̿�֪����Ӧ����Ψһ���������̼�ķų���ɷ�Ӧ��ʣ������Һ����������ȷ�Ӧǰ������2.2g������жϷų�������̼���������Ϊ2.2g��
��3���貹��Ʒ��̼��Ƶ�����Ϊx
CaCO
3+2HCl=CaCl
2+H
2O+CO
2��
100 44
x 2.2g
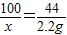
x=5g
�ʲ���Ʒ��̼��Ƶ�����������
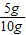
×100%=50%
��4��CaCO
3�и�Ԫ�ص���������=
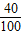
×100%=40%
����Ʒ�и�Ԫ�ص�ʵ�ʺ���=
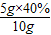
×100%=20%
�ʴ�Ϊ����1��CaCO
3+2HCl=CaCl
2+H
2O+CO
2������2��2.2g����3������Ʒ��̼��Ƶ�����������50%����4���ò���Ʒʵ�ʺ�����Ϊ20%��
���������ݷ�Ӧ�Ļ�ѧ����ʽ�ܱ�ʾ��Ӧ�и����ʵ�������ϵ���ɷ�Ӧ��ij���ʵ������ɼ������Ӧ���������ʵ�������

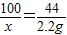
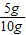 ×100%=50%
×100%=50%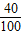 ×100%=40%
×100%=40%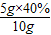 ×100%=20%
×100%=20%

 �Ƹ�С״Ԫ�������������ϵ�д�
�Ƹ�С״Ԫ�������������ϵ�д� ����һ������ܼƻ�ϵ�д�
����һ������ܼƻ�ϵ�д�