
��9�֣�����ʵ��Ҫ��������
��һ����ͼ��ʵ������ȡ�����һ��װ��ͼ����Ҫ��ش����⣺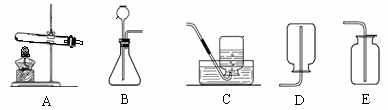
��1��ʵ�������Ը������Ϊԭ����װ��A����ȡ��������װ��A��Ҫ�Ľ��ĵط��� ����Ӧ�Ļ�ѧ����ʽ�� ��
��2�����װ��A�����Եķ����� ��
��3��ij����ȿ�����װ��C�ռ��ֿ�����Dװ���ռ������������е�����������
�����еĻ�ѧ������ ��
������ij��ȤС������ͼ��ʾװ���ռ���������Ӧ���������弰����������ø�װ����ȡ������ͨ����Һ©���м���30mLϡ���ᣬ��ƽ����ƿ��ʢ�ŵ�0.65gп����ַ�Ӧ���������������ش��������⣺
��4������ͼװ�����ӳ�����ʵ��װ�ã�����˳������Ϊa �� �� ��d����ӿ���ĸ����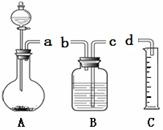
��5��������0.65gп������ϡ���ᷴӦ�ڸ�������Ӧ���ռ���224mL����������3��ʵ���ռ������������ƽ��ֵԼΪ242mL�����������Ҫԭ���� �����ţ���
A.�ռ������������������ B. �ռ�����������˿����������
��6��Ϊ�˼�Сʵ������ȡ�������ѡ�õ����� ��
 ��������һ���þ�ϵ�д�
��������һ���þ�ϵ�д� Сѧ��10����Ӧ����ϵ�д�
Сѧ��10����Ӧ����ϵ�д�
| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
| ||
| ||
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
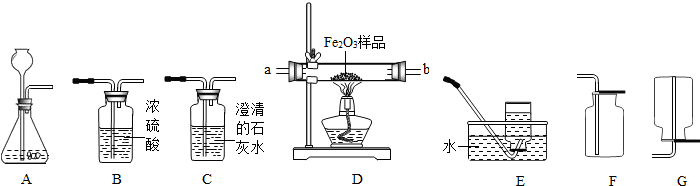
��6�֣���ͼ��ʾΪʵ�����г����������Ʊ����������ռ�������ʵ��IJ����������Ը�����ĿҪ�ش��������⣺

��1�����Թ���������ҺΪԭ�ϣ�������������������ʵ�������Ʊ����ռ������������
����ѡ����������˳��ΪA �� �� ��
������A�з����Ļ�ѧ����ʽ�� ��
��2�����ú�����CO2��CO����ⶨij����Fe2O3��Ʒ�Ĵ��ȣ����ʲ�������������������˳��Ϊ����������C1��B��D��C2��C1�� C2Ϊ����ʢ�г���ʯ��ˮ��������
��C2�е�����Ϊ ����ѧ����ʽΪ ��
��ͨ���ⶨC2�����ı仯������Fe2O3��Ʒ�Ĵ��ȣ���������C1���������Ľ��
�� ���ƫ��ƫС��������Ӱ�족����
�۴ӻ����ĽǶȷ�������װ����ƵIJ���֮�� ��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ��2012��ɽ��ʡ������������һģ��ѧ�Ծ����������� ���ͣ������
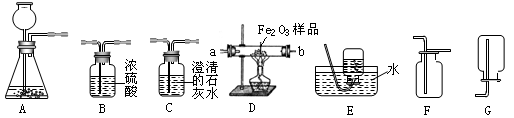
��6�֣���ͼ��ʾΪʵ�����г����������Ʊ����������ռ�������ʵ��IJ����������Ը�����ĿҪ�ش��������⣺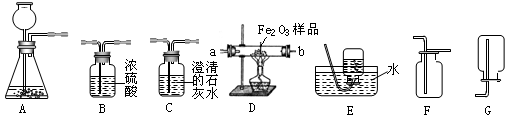
��1�����Թ���������ҺΪԭ�ϣ�������������������ʵ�������Ʊ����ռ������������
����ѡ����������˳��ΪA �� �� ��
������A�з����Ļ�ѧ����ʽ�� ��
��2�����ú�����CO2��CO����ⶨij����Fe2O3��Ʒ�Ĵ��ȣ����ʲ�������������������˳��Ϊ����������C1��B��D��C2��C1�� C2Ϊ����ʢ�г���ʯ��ˮ��������
��C2�е�����Ϊ ����ѧ����ʽΪ ��
��ͨ���ⶨC2�����ı仯������Fe2O3��Ʒ�Ĵ��ȣ���������C1���������Ľ��
�� ���ƫ��ƫС��������Ӱ�족����
�۴ӻ����ĽǶȷ�������װ����ƵIJ���֮�� ��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ��2012�콭��ʡ�Ͼ��й�¥�����꼶�п���ģ��ѧ�Ծ����������� ���ͣ������
��7�֣���ͼ��ʾΪʵ�����г����������Ʊ����������ռ�������ʵ��IJ����������Ը�����ĿҪ�ش��������⣺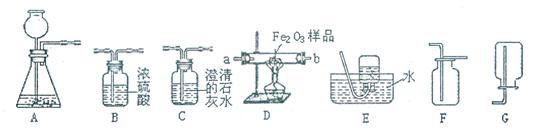
��1�����Թ���������ҺΪԭ�ϣ�������������������ʵ�������Ʊ����ռ������������
����ѡ����������˳��ΪA �� �� ��
������A�з�����Ӧ�Ļ�ѧ����ʽ�� ��
�ۼ��������Ѿ������ķ����� ��
��2�����ú�����CO2��CO����ⶨij����Fe2O3��Ʒ�Ĵ��ȣ����ʲ���������װ�õ�����˳��Ϊ����������C1��B��D��C2��C1�� C2Ϊ����ʢ�г���ʯ��ˮ��װ�ã�
��C2�е�����Ϊ ��
��ͨ���ⶨC2�����ı仯������Fe2O3��Ʒ�Ĵ��ȣ���������C1���������Ľ���� ���ƫ��ƫС��������Ӱ�족����
�۴ӻ����ĽǶȷ�������װ����ƵIJ���֮�� ��
�鿴�𰸺ͽ���>>
����ѧУ��ѡ - ��ϰ���б� - �����б�
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com