
Ŀǰ������60%��þ�ǴӺ�ˮ����ȡ�ġ���ˮ��þ����Ҫ��������:
��ش���������:
(1)�����ӷ�Ӧ�ĽǶ�˼��,�ں�ˮ�м���ʯ�������������������,д���ڳ������з�����Ӧ�����ӷ���ʽ����������������������������
(2)ʯ��������ʯ����ˮ�γɵĻ�����,�ӳ�����ú���ѧ��Դ,��߾���Ч��ĽǶ�,������ʯ�ҵ���Ҫԭ����Դ�ں����е�����������
(3)����A����������,����B������������
(4)����������Լ�a����������(�ѧʽ)��
(5)��ˮMgCl2������״̬��,ͨ�������Mg��Cl2,�÷�Ӧ�Ļ�ѧ����ʽΪ���������������ӿ��dzɱ��ͷ���ѭ�����õĽǶ�,����������������������������
(6)��ˮ��þ�Ĺ���,ΪʲôҪ����ˮ�е��Ȼ�þת��Ϊ������þ,��ת��Ϊ�Ȼ�þ?
_____________________________________________
 ��У����ϵ�д�
��У����ϵ�д�
| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ���ѡ��
�����й�˵����ȷ����
| A�����¼�������þ��̼�Ļ��������Ƶ���þ |
| B������ұ��������ͨ���û���Ӧ�õ������� |
| C����ˮ����Ĺ����в�����������ԭ��Ӧ |
| D�����õ��ķ������ԴӺ�ˮ�л�õ�ˮ |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
����ѧ����ѡ��2����ѧ�뼼������15�֣�
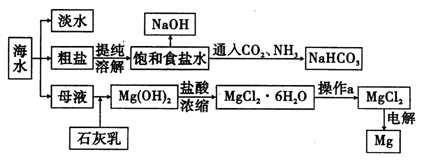
ͨ����ˮ�ܻ�õ�ˮ��ʳ�Ρ�þ�ȣ�ʳ�οɽ�һ�������ȼҵ����ش��������⡣
��1���о����ֺ�ˮ�����ķ�����_________��_________��
��2���ȼҵͨ����ⱥ��ʳ��ˮ�ܻ���ռ�����������ʣ��÷�Ӧ�Ļ�ѧ����ʽΪ_____________����ͼ�������ӽ���Ĥ����ⱥ��ʳ��ˮ��ԭ��ʾ��ͼ������ʯī�ӵ�Դ_________�������ʱ���缫�ĵ缫��ӦʽΪ_________����������ͨ�����ӽ���Ĥ����Ҫ������__________��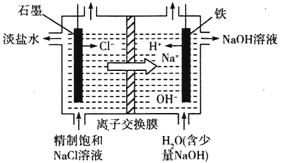
��3�������Ƽ�У�������ʳ��ˮ��ͨ��CO2��NH3�Ʊ�NaHCO3���仯ѧ����ʽΪ____________����ͨ�����__________���ѧʽ������������__________________ ��������NaHCO3���ȷֽ���Ʊ����
��4��Ŀǰ������60%���ϵ�þ���ǴӺ�ˮ����ȡ�ģ���֪��MgO��MgCl2���۵�ֱ�Ϊ2852���714�档����˵����ҵ�ϲ��õ������MgCl2�����ǵ������MgO������__________ ______________________________________________________________________ ��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
��ѡ��2����ѧ�뼼������15�֣�
�������������գ���Ŀǰ����ˮ���塱������Ҫ����֮һ���乤���������£�
��1������ܵ����ӷ�Ӧ����ʽΪ ��
��2��������ˮ���������������������õ�Һ������ˮ�Ļ������ǵ�����ܶ����ϴ����Һ���ʵ���ҷ���Ϊ ��
��3������������ʵ�����н��У����õ��IJ��������� ��
��4����ҵ������ֱ��������ĺ�ˮ�õ�Һ�壬��Ҫ����������������SO2���ա��Ȼ�����ԭ���� ��
��5����±ˮ������������ȡ����þ���û�ѧ����ʽ��ʾ�ӿ�±ˮ��ȡ����þ�ķ�Ӧԭ�� ��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
��ˮ��þ���ܴ���ԼΪ2.1��1015t��Ŀǰ������60%��þ���Ժ�ˮ����ҵ��ģ��ˮ��þ��������ͼ��ʾ��
�Իش��������⣺
(1)��ˮ��þ�Ĺ����Т١��ڷ�Ӧ�Ļ�ѧ����ʽ��
��_____________________________________��
��______________________________________��
(2)���Ȼ�þ��Һ��ȡ��ˮ�Ȼ�þ���壬�����������____________________
(3)Ϊ��ʹMgSO4��ȫת��ΪMg(OH)2����������ʯ��Ҫ������Ȼ������Mg(OH)2����������Ca(OH)2�ܽ�ȣ�Ӧ����________�����롣
(4)����þ�����Ĺ�ҵұ��������������֮�������в�֮ͬ���±�������þ���Ȼ�þ���۷е����ݣ�
| ���� | ����þ | �Ȼ�þ |
| �۵�(��) | 2 852 | 714 |
| �е�(��) | 3 600 | 1 412 |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
������˵������ȷ����____ ��
| A����������ijЩ���ַ����������� |
| B����������Ӳ�ȵ�ˮ��Ҫ�ü��ȵķ������������� |
| C�����Ṥҵ�У��ڽӴ��Ұ�װ�Ƚ�������Ϊ������S03ת��ΪH2S04ʱ�ų������� |
| D���ϳɰ���ҵԭ��������ʱ������̼�����Һ���ճ�ȥ������̼ |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
��������к���Cr(OH)3��Al2O3��ZnO��CuO��NiO������,��ҵ��ͨ�������±��ա���������������Na2Cr2O7�����ʡ�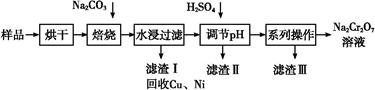
��֪:��Na2CrO4��Һ�к�������NaAlO2��Na2ZnO2������
(1)ˮ�������Һ������������(��ᡱ��������С�)��
(2)����������չ���������Na2CrO4�Ļ�ѧ����ʽ��
��������Cr(OH)3+��������Na2CO3+������������ ��������Na2CrO4+��������CO2+������������
��������Na2CrO4+��������CO2+������������
(3)���������Ҫ�ɷ���Zn(OH)2������������
(4)��ϵ�в�������Ϊ:��������H2SO4,��������,��ȴ�ᾧ,���ˡ���������H2SO4Ŀ������ ��
��֪:�ٳ�ȥ����II��,��Һ�д������·�Ӧ:
2CrO42��+2H+ Cr2O72��+H2O
Cr2O72��+H2O
��Na2Cr2O7��Na2CrO4�ڲ�ͬ�¶��µ��ܽ�����±�
| �¶� �ܽ�� ��ѧʽ | 20 �� | 60 �� | 100 �� |
| Na2SO4 | 19.5 | 45.3 | 42.5 |
| Na2Cr2O7 | 183 | 269 | 415 |
| Na2CrO4 | 84 | 115 | 126 |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ���ѡ��
��������������,��Ŀǰ����ˮ���塱������Ҫ����֮һ���乤����������,���в���ȷ����(����)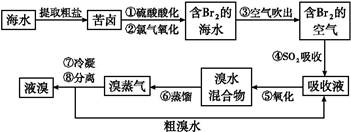
A.��������������ữ�ɷ�ֹCl2��Br2��ˮ��Ӧ
B����ܵ����ӷ���ʽBr2+SO2+H2O 2H++2Br-+S
2H++2Br-+S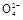
C�������ڱ���λ��4���ڢ�A�塣
D.�������������������õ�Һ������ˮ�Ļ������÷�Һ©��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
�㶫ʡ���ŷḻ�ĺ�����Դ����ˮ��ȡʳ�κ�Br2�Ժ����±���������Ʊ�������MgCl2��MgO����±�к���Mg2����Cl��������������Na����Fe2����Fe3����CO(NH2)2�ȡ��Ʊ�������ͼ��ʾ��
(1)�����ijɷ���______(�ѧʽ)����Һ������������Ҫ�������� (д���ӷ���)��
(2)��NaClO��ȥ����CO(NH2)2ʱ������������⣬�����ܲ������ѭ�������ʣ���÷�Ӧ�Ļ�ѧ����ʽΪ________������NaClO������������______��
(3)ֱ�ӽ�MgCl2��6H2O��ǿ���ܵõ�MgO����Ӧ�Ļ�ѧ����ʽ��_____����MgCl2��6H2O�Ʊ���ˮMgCl2�����У�����Ҫ�Ļ�ѧ�Լ���________��
(4)��ˮ������������ճ�ʪ�����е�Br2������SO2���壬SO2����Br2�����ӷ���ʽ��_________________________________��SO2�������Դ�����Ṥҵ��β����ͬʱ��SO2β��Ҳ���ð�ˮ���գ���Ϊ�Ʊ����ʵ�ԭ�ϣ�SO2�����ð�ˮ���յõ��IJ��������________��
�鿴�𰸺ͽ���>>
����ѧУ��ѡ - ��ϰ���б� - �����б�
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com