
 ��2011?�㶫�����ù��ܺ�������ɽ�CO2��H2O��g��ת��ΪCH4��O2�����������ʱ���ڲ�ͬ������I��II��III�������£�CH4���������ʱ��ı仯��ͼ��ʾ��
��2011?�㶫�����ù��ܺ�������ɽ�CO2��H2O��g��ת��ΪCH4��O2�����������ʱ���ڲ�ͬ������I��II��III�������£�CH4���������ʱ��ı仯��ͼ��ʾ��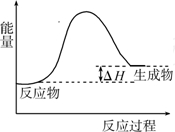 ��
��
| c(CO)c3(H2) |
| c(CH4)c(H2O) |
| 0.10��0.303 |
| (x-0.10)2 |
| 0.10mol |
| 0��11mol |



| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ��Ķ�����
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
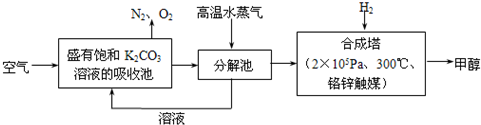
(8��)(2011���㶫ģ��)��֪һ��̼ԭ����ͬʱ���������ǻ�ʱ����������ת����
�������ͼ�ش�
(1)E�к��еĹ����ŵ�������________���۵ķ�Ӧ������________��C�����Ƶ�������ͭ��Һ��Ӧ�Ļ�ѧ����ʽΪ_______________________________________��
(2)��֪B����Է�������Ϊ162����ȼ�ղ�����n(CO2)��n(H2O)��2��1��F�ǵ���оƬ�еĸ߷��ӹ��������Ҫԭ�ϣ�F���ص��ǣ�������FeCl3��Һ������ɫ��Ӧ�����ܷ����Ӿ۷�Ӧ���۱����ϵ�һ��ȡ����ֻ�����֡�F��һ�������·����Ӿ۷�Ӧ�Ļ�ѧ����ʽΪ________��
(3)������G��F��ͬ���칹�壬�����ڷ����廯����ܷ���������Ӧ��G������________�ֽṹ��д����������һ��ͬ���칹��Ľṹ��ʽ_____________________��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ��2012��߿���ѧһ�ָ�ϰ�����������ר���ۺϲ��ԣ��ս̰棩 ���ͣ������
(8��)(2011���㶫ģ��)��֪һ��̼ԭ����ͬʱ���������ǻ�ʱ����������ת����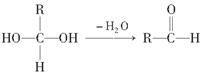
�������ͼ�ش�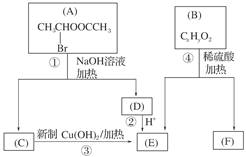
(1)E�к��еĹ����ŵ�������________���۵ķ�Ӧ������________��C�����Ƶ�������ͭ��Һ��Ӧ�Ļ�ѧ����ʽΪ_______________________________________��
(2)��֪B����Է�������Ϊ162����ȼ�ղ�����n(CO2) ��n(H2O)��2 ��1��F�ǵ���оƬ�еĸ߷��ӹ��������Ҫԭ�ϣ�F���ص��ǣ�������FeCl3��Һ������ɫ��Ӧ�����ܷ����Ӿ۷�Ӧ���۱����ϵ�һ��ȡ����ֻ�����֡�F��һ�������·����Ӿ۷�Ӧ�Ļ�ѧ����ʽΪ________��
(3)������G��F��ͬ���칹�壬�����ڷ����廯����ܷ���������Ӧ��G������________�ֽṹ��д����������һ��ͬ���칹��Ľṹ��ʽ_____________________��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ��2011-2012ѧ��߿���ѧһ�ָ�ϰ�����������ר���ۺϲ��ԣ��ս̰棩 ���ͣ������
(8��)(2011���㶫ģ��)��֪һ��̼ԭ����ͬʱ���������ǻ�ʱ����������ת����

�������ͼ�ش�
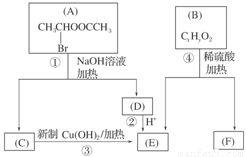
(1)E�к��еĹ����ŵ�������________���۵ķ�Ӧ������________��C�����Ƶ�������ͭ��Һ��Ӧ�Ļ�ѧ����ʽΪ_______________________________________��
(2)��֪B����Է�������Ϊ162����ȼ�ղ�����n(CO2) ��n(H2O)��2 ��1��F�ǵ���оƬ�еĸ߷��ӹ��������Ҫԭ�ϣ�F���ص��ǣ�������FeCl3��Һ������ɫ��Ӧ�����ܷ����Ӿ۷�Ӧ���۱����ϵ�һ��ȡ����ֻ�����֡�F��һ�������·����Ӿ۷�Ӧ�Ļ�ѧ����ʽΪ________��
(3)������G��F��ͬ���칹�壬�����ڷ����廯����ܷ���������Ӧ��G������________�ֽṹ��д����������һ��ͬ���칹��Ľṹ��ʽ_____________________��
�鿴�𰸺ͽ���>>
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com