
��֪��������ͨ����������S8(б����)����ʽ���ڣ���������״̬ʱ������S2��S4��S6��S8�ȶ���ͬ�������壬����S4��S6��S8�������ƵĽṹ�ص㣬��ṹ����ͼ��ʾ��
��һ�������£�S8(s)��O2(g)������Ӧ����ת��ΪSO2(g)��SO3(g)����Ӧ���̺�������ϵ������ͼ��ʾ(ͼ�еĦ�H��ʾ����1 mol���������)��
��1��д����ʾS8ȼ���ȵ��Ȼ�ѧ����ʽ___________________________________��
��2��д��SO3�ֽ�����SO2��O2���Ȼ�ѧ����ʽ_______________________________________________________________��
��3����ѧ�Ϲ涨�����γ�1 mol��ѧ�����ջ�ų���������Ϊ�û�ѧ���ļ��ܣ���λkJ��mol������֪�������ļ���Ϊd kJ��mol��1���������ļ���Ϊe kJ��mol��1����S8������������ļ���Ϊ____________________________________��

| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
���õ�����������һ�����������ɰ�����һ���淴Ӧ���ϳɰ�����һ����Ҫ�Ļ�����Ӧ������������Һ���Ͱ�ˮ��
���������գ�
��1����ͼ��ʾ�ϳɰ�ʱ����1mol������ʱ�������仯��E�ĵ�λΪkJ����д���ϳɰ����Ȼ�ѧ����ʽ____________________��
��������E1��E2��E3��ʾ������ͼ�е�ʵ�������߲�����ʲô��Ӧ���������˱仯��
��2����һ���¶��£�����4a mol H2��2amol N2����VL���ܱ������У�5���Ӻ���N2��ת����Ϊ50%����ö�ʱ����H2��ʾ�ķ�Ӧ����Ϊ__________Ħ��/(��?��)������ʱ�����������Ͷ��a mol H2��amol N2��2amol NH3���ж�ƽ���ƶ��ķ�����_____��������ƶ����������ƶ������ƶ�����
��3��Һ����ˮ���ƣ�Ҳ�ܵ��룺2NH3 NH4++ NH2����ij�¶�ʱ�������ӻ�K=2��l0-30�����¶��£��ٽ�����NH4Cl�������Һ���У�K____________2��10-30�����������������=�������ڽ�����������Ͷ��Һ���У���ȫ��Ӧ��������Һ�и�����Ũ�ȴ�С��ϵΪ��_______
NH4++ NH2����ij�¶�ʱ�������ӻ�K=2��l0-30�����¶��£��ٽ�����NH4Cl�������Һ���У�K____________2��10-30�����������������=�������ڽ�����������Ͷ��Һ���У���ȫ��Ӧ��������Һ�и�����Ũ�ȴ�С��ϵΪ��_______
��4�����������İ�ˮ������ʱ��Ҫϡ�͡���ˮϡ��0��1mol/Lϡ��ˮʱ����Һ������ˮ�������Ӷ����ٵ���
| A��c(NH4+)/c(NH3?H2O) | B��c(NH3?H2O)/c(OH-) |
| C��c(H+)/c(NH4+) | D��c(OH-)/c(H+) |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
��һ��С�ձ������Լ20 g����ĥ�ɷ�ĩ��������������[Ba(OH)2��8H2O]����С�ձ����������ѵ���3��4��ˮ�IJ���Ƭ�ϣ�Ȼ�����ձ��ڼ���Լ10 g�Ȼ�茶��壬�������ò�����Ѹ�ٽ��衣�Իش��������⣺
(1)д����Ӧ�Ļ�ѧ����ʽ�� _____________________________��
(2)ʵ����Ҫ�����ò�����Ѹ�ٽ����ԭ����_____________________________��
(3)���ʵ����û�п���������������ܵ�ԭ����(�����������������ԭ��)__________________________ _____________��
(4)���û�п�����������������ǻ����Բ�ȡ��Щ��ʽ��˵���÷�Ӧ���ȣ�
_______________________________________(������ַ���)��
(5)�����������˵���÷�Ӧ��һ��________(��ų��������ա�)�����ķ�Ӧ�����Ͽ��ɻ�ѧ��________(����ա��ų���)������________(�>����<��)�γ��»�ѧ��________(����ա��ų���)��������
(6)�÷�Ӧ�ڳ����¾Ϳɽ��У�˵��_________________________________��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
ǰ��ʱ��ϯ���ҹ����������������ǵ�������������˼����Ӱ�죬��ͳ���ҹ����ֳ���������ռȫ��һ�룬����������PM2.5ϸ���Ӱ���(NH4)2SO4��NH4NO3������������л������P�ﳾ�ȡ�
��1���л�������IJ�����Ҫ�����ڲ���ȫȼ�յ��µ�����Ȼ�ѧ����ʽ���£�
��C(s)��O2(g)=CO2(g)����H1����94kJ��mol��1��
��C8H16(l)+12O2(g)=8CO2(g)+8H2O(l) ��H2����1124kJ��mol��1
��C8H16(l)+4O2=8C��g��+8H2O��l����H3�� kJ��mol��1
��2�����������ѿɹ��ӷ����л���Ⱦ�VOCs��������ˮ�������ڣ�������ϩ���ⷴӦΪ��C2HCl3+2O2��2CO2+HCl+Cl2�������㹻���Ľ�����β����ʵ���Ҽ���������������ļ����ǣ� ��ͨ�������Ƿ��ֻ��ж��ָ��������֮һΪ��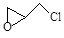 ������л���˴Ź��������� �� ���塣
������л���˴Ź��������� �� ���塣
��֪��Cu(OH)2�Ƕ�Ԫ��������ᣨH3PO3���Ƕ�Ԫ���ᣬ��NaOH��Һ��Ӧ������Na2HPO3��
��3����ͭ����Һ��Cu2������ˮ�ⷴӦ�����ӷ���ʽΪ____���÷�Ӧ��ƽ�ⳣ��Ϊ____������֪��25��ʱ��Ksp[Cu(OH)2]��2.0��10��20mol3/L3��
��4�����Na2HPO3��Һ�ɵõ������ᣬװ����ͼ��˵������Ĥֻ����������ͨ������Ĥֻ����������ͨ����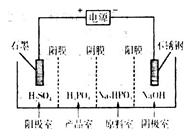
�������ĵ缫��ӦʽΪ____________________��
�ڲ�Ʒ���з�Ӧ�����ӷ���ʽΪ____________��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
�о�CO2�����öԴٽ���̼���Ĺ���������Ҫ�����塣
(1)��CO2�뽹̿��������CO��CO�����������ȡ�
��֪��Fe2O3(s)��3C(s)=2Fe(s)��3CO(g)����H1����489.0 kJ��mol��1
C(s)��CO2(g)=2CO(g)����H2����172.5 kJ��mol��1��
��CO��ԭFe2O3���Ȼ�ѧ����ʽΪ________________________________
(2)ijʵ�齫CO2��H2����һ��������ܱ������У������ֲ�ͬ�����·�����Ӧ��CO2(g)��3H2(g) CH3OH(g)��H2O(g)����H����49.0 kJ��mol��1�����CH3OH�����ʵ�����ʱ��ı仯��ͼ��ʾ����ش��������⣺
CH3OH(g)��H2O(g)����H����49.0 kJ��mol��1�����CH3OH�����ʵ�����ʱ��ı仯��ͼ��ʾ����ش��������⣺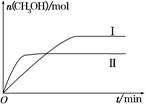
�ٸ÷�Ӧ��ƽ�ⳣ���ı���ʽΪK��________��
�����ߢ��Ӧ��ƽ�ⳣ����С��ϵΪK��________K��(����ڡ��������ڡ���С�ڡ�)��
������ͼa��b��c�����У�H2��ת�����ɸߵ��͵�˳����________(����ĸ)��
(3)�������������������£����������ѹ����ԭ����1/2����ԭƽ����ȣ������й�˵����ȷ����________(�����)��
a��������Ũ�ȼ�С
b������Ӧ���ʼӿ죬�淴Ӧ����Ҳ�ӿ�
c���״������ʵ�������
d������ƽ��ʱn(H2)/n(CH3OH)����
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
������Ϊһ������Դ�ڻ�ѧ����Ӧ�ù㷺����ش��������⡣
��1����¯ұ�������У������ڴ���Ӧ���в���ˮú����CO��H2����ԭ���������йط�ӦΪ��CH4��g����CO2��g��=2CO��g����2H2��g������H��260 kJ��mol��1
��֪��2CO��g����O2��g��=2CO2��g����H����566 kJ��mol��1��
��CH4��O2��Ӧ����CO��H2���Ȼ�ѧ����ʽΪ____________________________________��
��2������ͼ��ʾ��װ�â�Ϊ����ȼ�ϵ�أ��������ҺΪKOH��Һ����ͨ��װ�â�ʵ�������϶�ͭ��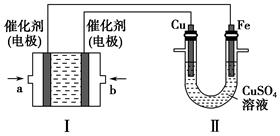
��a��Ӧͨ��________���CH4����O2������b���缫�Ϸ����ĵ缫��Ӧʽ��_________________________________________________________________��
�ڵ�ƽ�����װ�â�����Һ��pH________����д�������С�����䡱����ͬ����װ�â���Cu2�������ʵ���Ũ��________��
�۵�ƽ�����װ�â���Һ�е������ӳ���OH���������________������ˮ�⣩��
���ڴ˹���������ȫ��Ӧ��װ�â������������仯12.8 g����װ�â������������ļ���________L����״���£���
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
CH4��H2��C�������ʵ���Դ���ʣ�����ȼ�յ��Ȼ�ѧ����ʽΪ��
��CH4(g)��2O2(g)=CO2(g)��2H2O(l)����H����890.3 kJ��mol��1��
��2H2(g)��O2(g)=2H2O(l)����H����571.6 kJ��mol��1��
��C(s)��O2(g)=CO2(g)����H����393.5 kJ��mol��1��
(1)����д���һ�ּ���ϸ������������øʹ������O2���ò���������������ϸ��ʹ1 mol��������CO2������Һ̬ˮ���ų�������________(���������������)890.3 kJ��
(2)������CO2�����ںϳɺϳ���(��Ҫ�ɷ���һ����̼������)��CH4��CO2=2CO��2H2��1 g CH4��ȫ��Ӧ���ͷ�15.46 kJ����������
����ͼ�ܱ�ʾ�÷�Ӧ�����������仯����________(����ĸ)��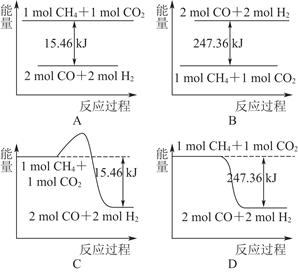
���������ʵ�����Ϊ1 mol��CH4��CO2����ij�����ܱ������У���ϵ�ų�����������ʱ��ı仯��ͼ��ʾ����CH4��ת����Ϊ________��
(3)C(s)��H2(g)����Ӧ������C(s)��2H2(g)=CH4(g)�ķ�Ӧ����ֱ�Ӳ�������ͨ��������Ӧ�������C(s)��2H2(g)=CH4(g)�ķ�Ӧ�Ȧ�H��________��
(4)Ŀǰ���������������ʵ��о���ȼ���о����ص㣬���й��������������ʵ��о������п��е���________(����ĸ)��
| A��Ѱ�����ʴ�����ʹCO2��H2O��Ӧ����CH4��O2�����ų����� |
| B��Ѱ�����ʴ������ڳ��³�ѹ��ʹCO2�ֽ�����̼��O2 |
| C��Ѱ�����ʴ���������̫����ʹ�����е�CO2�뺣���ɵ�CH4�ϳɺϳ���(CO��H2) |
| D������̬̼�ϳ�ΪC60����C60��Ϊȼ�� |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
��ĿǰΪֹ���ɻ�ѧ��ת��Ϊ���ܻ������Ȼ������ʹ������Ҫ����Դ��
��1����ѧ��Ӧ�зų������ܣ��ʱ䣬��H���뷴Ӧ����������ڷ�Ӧ�����жϼ����γ��¼����������պͷų������Ĵ�С�йء�
��֪��H2��g����Cl2��g��=2HCl��g�� ��H����185 kJ/mol������1 mol H��H�����յ�����Ϊ436 kJ������1 mol Cl��Cl�����յ�����Ϊ247 kJ�����γ�1 mol H��Cl���ų�������Ϊ ��
��2��ȼ��ȼ�ս��������Ļ�ѧ��ת��Ϊ��������Ҫ�����ܡ���֪��
��CH4��g����2O2��g��=CO2��g����2H2O��l�� ��H����890��3 kJ��mol-1
��C��s,ʯī����O2��g��=CO2��g�� ��H����393��5 kJ��mol��1
��2H2��g����O2��g��=2H2O��l�� ��H����571��6 kJ��mol-1
��״����22��4 L�����ͼ���Ļ�������������������г��ȼ�շ�Ӧ�ų�588��05 kJ��������ԭ��������������������� ���������������Ȼ�ѧ����ʽ������C��s,ʯī����2H2��g��=CH4��g���ķ�Ӧ�Ȧ�HΪ ��
�鿴�𰸺ͽ���>>
����ѧУ��ѡ - ��ϰ���б� - �����б�
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com