
����Ŀ���Ʊ���Ϊ�����еġ��ֽ����������
�������еĸƣ�
��1������ȱ�Ʋ��ᵼ��_____������ĸ����ͬ����
A ٪��֢ B ���Ͳ� C ��������
��2�������и�Ԫ����Ҫ�����ڹ����������У����ǻ������[Ca10��PO4��6��OH��2]��ʽ���ڣ������������PO4�����ϼ�Ϊ_____��
�����ʸƣ�
������⣺�ƵĻ�����ܶ࣬Ϊʲô���Ǻ��������ʸ��أ�
˼�����룺�ƵĻ�ѧ���ʻ��á�
ʵ����֤һ��

�������ϣ���һ����ѧ��Ӧ�У���Ԫ�ػ��ϼ����ߣ�ͬʱ����Ԫ�ػ��ϼ۽��͡�
��3��ʵ����ۣ���д������ˮ��Ӧ�Ļ�ѧ����ʽ_____��
���÷�Ӧ_____������ա��ų�����������
��������еĸƣ�
������⣺һ����ʯ�Ҹ������ʹ��һ��ʱ����������Щ���壿
������裺�����к��������ơ��������ơ�̼��ơ�
ʵ����֤����
��4��ȡ������Ʒ����ˮ�У����¶��������ߣ�����_____���μӷ�̪��Һ���죻����������ϡ���ᣬ��۲쵽_____��������к���̼��ơ�
��5��С����Ϊ���ж��Ƿ����������ƣ�������_____��
��6��Ϊ�˽�һ��ȷ��������Ʒ�ijɷּ�������ʵ��С��������ͼ��ʾ��װ�ã��г�����ʡ�ԣ�����ʵ�顣

�������ϣ��������Ƽ�����580�桢̼��Ƽ�����825��ֱ�ֽ��������������
��ʵ��ǰ����Ҫ_____����װ��ҩƷ��
��װ��E��������_____��
�����µ������ʹװ��B�з�Ӧ��ȫ����ͨһ��ʱ���������÷�Ӧǰ��װ��C��D�ֱ�����9.0g��11.0g����װ��B�з�Ӧ��ʣ��Ĺ����������������ˮ���ܽ⣬�������ù��壨����ˮ������18.0g����ԭ������Ʒ�и��ɷֵ�����������д��������̣�_____��
���𰸡�A -3 Ca+2H2O=Ca��OH��2+H2�� �ų� ������ �����ݲ��� ��������ˮ��Ӧ���ɵ�����������ҺҲ�ʼ��� ���װ�õ������� ��ֹ�����е�CO2��H2O����D ԭ������Ʒ�к��������Ƶ�����Ϊ37g����̼��Ƶ�����Ϊ25g���������Ƶ�����Ϊ14g��
��������
��1������ȱ�ƻᵼ�����Ͳ��������ɣ����ᵼ��٪��֢����ѡ��A��
��2����Ԫ����+2�ۣ���Ԫ����-2�ۣ���������-1�ۣ���������Ļ��ϼ���x�������ڻ��������������ϼ۴�����Ϊ�㣬�ɵã���+2����10+6x+��-1����2=0����x=-3�ۣ����-3��
��3���ٸ���ˮ��Ӧ�Ļ�ѧ����ʽ��Ca+2H2O=Ca��OH��2+H2�������Ca+2H2O=Ca��OH��2+H2����
�ڿ�ʼʱ��Һ����ǣ����º��ɳ�����Һ������Ϊ���������ܽ�������¶����߶���С����˸÷�Ӧ���ڷ��ȷ�Ӧ������ų���
��4��ȡ������Ʒ����ˮ�У����¶��������ߣ����������ƣ��������ɵ�����������Һ�Լ��ԣ���˵μӷ�̪��Һ���죻����������ϡ���ᣬ��۲쵽�����ݲ���������庬��̼��ƣ���������ƣ������ݲ�����
��5����������ˮ��Ӧ���ɵ�����������ҺҲ�ʼ��ԣ�������ж��Ƿ����������ƣ������������ˮ��Ӧ���ɵ�����������ҺҲ�ʼ��ԣ�
��6����ʵ��ǰ����Ҫ���װ�õ������ԣ���װ����Ʒ��������װ�õ������ԣ�
��װ��E��Ϊ�˷�ֹ�����еĶ�����̼��ˮ��������װ��D��Ӱ��ʵ�����������ֹ�����е�CO2��H2O����D��
�ۿ��¼���ʹװ��B�з�Ӧ��ȫ����ͨһ��ʱ���������÷�Ӧǰ��װ��C��D�ֱ�����9.0g��11.0g�����������Ʒֽ�����ˮ������Ϊ9.0g��̼��Ʒֽ����ɶ�����̼������Ϊ11.0g�����������Ƶ�����Ϊx���ֽ����ɵ������Ƶ�����Ϊy��̼��Ƶ�����Ϊz���ֽ����ɵ������Ƶ�����Ϊm����

��װ��B�з�Ӧ��ʣ��Ĺ����������������ˮ���ܽ⣬�������ù��壨����ˮ������18g����μӷ�Ӧ�������Ƶ�����Ϊn����
��ԭ������Ʒ��CaO������Ϊ56g-28g-14g=14g
�����Ϸ�����֪��ԭ������Ʒ�к��������Ƶ�����Ϊ37g����̼��Ƶ�����Ϊ25g���������Ƶ�����Ϊ14g��
��ԭ������Ʒ�к��������Ƶ�����Ϊ37g����̼��Ƶ�����Ϊ25g���������Ƶ�����Ϊ14g��


 ��ʦ������Ԫ��ĩ���100��ϵ�д�
��ʦ������Ԫ��ĩ���100��ϵ�д� ��У������Ԫͬ��ѵ��������ϵ�д�
��У������Ԫͬ��ѵ��������ϵ�д�
| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
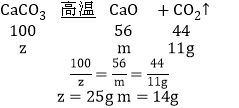
����Ŀ���������ͼ��ʾ��ʵ��װ��ͼ��ա�
(1)�����ٵ�������_________��
(2)ʵ������ A װ����ȡ�������÷�Ӧ�Ļ�ѧ����ʽΪ_____����Ҫ�ռ���Ϊ�������������ռ�װ��Ӧѡ��______(�����)����ͼH�������ȵ���˿����ʢ�������ļ���ƿ�У���ѧ����ʽΪ____��
(3)ʵ������ȡ������̼��ѡ�õķ���װ����_______(�����)���÷�Ӧ�Ļ�ѧ����ʽΪ_______ ����װ�õ��ŵ���_______���ռ�װ����ѡ E�����ſ������ռ����壬������Ӧ��_____(����a������b��)�˽��롣��Ӧ������Ҫ�ӷ�Ӧ��Ļ�����еõ��Ȼ��ƹ�����Ҫ�õ�������_______װ��(�����)��
(4)��ͼ�Ƕ�����̼�Ʊ������ʵ���ʵ��װ��ͼ��
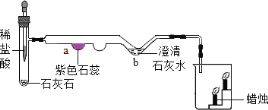
�Թ��з�Ӧ������a����ʵ��������_____________��b���Ļ�ѧ����ʽ��____________���ձ��е�ʵ��������______________��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
����Ŀ��ˮ��һ����������������ɳɷ֣�������밮ϧˮ��
��1���������ˮ�к���������Ϊˮ���ܽ���һ�������______��
��2�������У�����______����������ˮ��Ӳˮ������ˮ��
��3��������Ա�ø�ѹˮǹ����ԭ����Ҫ��______��
��4��������ʾ��ͼ�У�����ˮ�����ӿ���______![]() �����
�����![]() ��ʾ��
��ʾ��

��5��ˮ����Ȼ�����в��ֽ⣬����ͨ��������¿��Էֽ⣬�䷴Ӧ�Ļ�ѧ����ʽΪ______��
����ͼ��ʾ��װ�ý��е��ˮʵ�飬��A��B���ܼ���ˮ��رջ�����ͨ��һ��ʱ��ɹ۲쵽C�е�����Ϊ______��

��6���⻯�ƹ��峣���ڵ�ɽ��Ա����Դ�ṩ��������ˮ��Ӧ�����������ƺ���������Ӧ�Ļ�ѧ����ʽΪ______��
��7����ʵ������У�ˮҲ����������Ҫ�����ã��ֱ�д������ʵ����ˮ�����ã�
����˿�������е�ȼ��ʵ���У�����ƿ�ײ�������ˮ������______��
�����������е�ȼ��ʵ���У�����ƿ�ײ�������ˮ������______��
����ȡ��������ˮ���ռ�����ʱ������ƿ��ˮ������______��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
����Ŀ������ͼ1��ͼ2�����ֹ������ʵ��ܽ�����ش����⣨ˮΪ�ܼ�����

��1������ͼ1��P��ĺ�����_____________________��ȡһ��������A�ı�����Һ������Һ�¶ȴ�t�����͵�10������Һ�����ʵ���������__�����������С�����䡱����
��2������ͼ2��ҪʹC�ı�����Һ��ɲ�������Һ���ɲ��õķ����ǣ�_____�ͼ�ˮ��
��3��10��ʱ��A��B��C�ı�����Һ�����ʵ����������ɴ�С��˳����_____��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
����Ŀ������ˮ���������г���������ת����
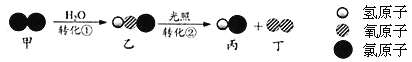
����˵����ȷ����
A. ��Ӧǰ����Ӳ��ɷ�B. ת�����б��Ͷ��ķ��Ӹ���֮��Ϊ1�U1
C. ת�����ǻ��Ϸ�ӦD. ת���١�������Ԫ�صĻ��ϼ۶������仯
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
����Ŀ����ͼA��B�ֱ���Ԫ����Ԫ�����ڱ����ṩ�IJ�����Ϣ��ԭ�ӽṹʾ��ͼ����ͼ�����ó������н����д������:

A. ���ݵ����Ų�����Ԫ�������ƻ�ѧ����
B. ԭ��A�ڻ�ѧ��Ӧ����ʧ���ӳ�������
C. ԭ��A�ĺ˵�����ͺ�����������Ϊ12
D. Ԫ��B��ɵĵ��ʾ����ȶ��Ļ�ѧ����
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
����Ŀ���������侭�á������������ǹ㷺ʹ�ã���ͼ�Ǵ����Ľṹͼ��ʹ�õ�ȼ�϶���(C4H10)��һ����ȼ����ɫ������Һ�������塣
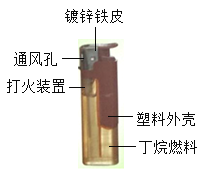
(1)���������п���������⣬��������������__________�����ö���ֹ�����⣬������Ҫ�ɷֵĻ�ѧʽ��______________��
(2)ʹ�ô���ʱ���������´��װ���ϵİ�ť�����װ�ò�����������ȼ���ӷ��ӵĽǶȽ��Ͷ���Һ��������������ԭ����____________________________________.
(3)��������ͨ��ף�˵��ȼ�յ�����֮��_________________________�������������г��ȼ�յ���������_______________��_________________________.
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
����Ŀ����7�֣���ͼ�ǹ������ذ�Ī���ֽ���ʹ��˵����IJ������ݣ�����˵������㣺

��1����Ī������Է���������____ ��
��2����Ī������̼Ԫ�ء���Ԫ�ء���Ԫ�ص���������____
��3����Ī��������Ԫ�ص�������������ȷ��0.1%����
��4�����һ������25kg�Ķ�ͯ������Ҫ���ð�Ī���֣�ÿ��Ҫ������������Ҫ�м�����̣�
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
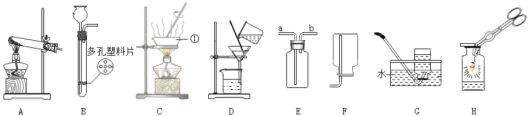
����Ŀ����ͼ��ij��ѧ��ȤС�顣��ƵĽ������˳���̽��ʵ�顣
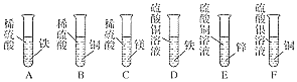
(1)����ʵ���У����������ɵ���________(��дװ�����)��
(2)�Թ�F�г��ֵ�ʵ��������______________��
(3)����ʵ���ܷ�֤��þ��п������ͭ�������ֽ����Ļ��ǿ����____��˵������_____��
(4)�ҹ��Ŵ���ʪ����ͭ�������������Թ�D�еķ�Ӧԭ������д���÷�Ӧ�Ļ�ѧ����ʽ��______��
�鿴�𰸺ͽ���>>
����ѧУ��ѡ - ��ϰ���б� - �����б�
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com